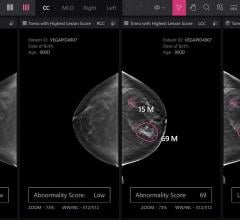
Ambra Health and St. Luke's University Health Network recently announced a new partnership that offers patients a streamlined method of image viewing and exchange.
November 18, 2016 — Vital Images Inc. announced that it will launch Vitrea Data Stream and the latest release of Vitrea ...
Accuray recently unveiled the company’s newest innovation, the Radixact Treatment Delivery System, a uniquely smart, fully integrated treatment delivery, treatment planning and data management system.
AT A GLANCE Organization: Expert Radiology Management Services, LLC Specialty: Subspecialty teleradiology — neuro and ...
Agfa HealthCare announced that it will introduce its new DR 800 X-ray room with Dynamic MUSICA at the 2016 annual meeting of the Radiological Society of North America (RSNA 2016). The multi-purpose solution covers radiography, fluoroscopy and advanced clinical applications.
HealthMyne has partnered with Gilda’s Club of Madison (Wis.) in an effort to make a difference in the lives of children and adults living with cancer during the 2016 Radiological Society of North America (RSNA) annual meeting.
Carestream will demonstrate the benefits offered by its Unified Core, which delivers a modular, scalable enterprise image management architecture, at the 2016 Radiological Society of North America (RSNA) conference, Nov. 27-Dec. 2 in Chicago.
Radiology departments have many different needs and face a wide variety of challenges that can impact their departments ...
November 17, 2016 — TeraRecon announced the release of an advanced integration with Agfa Healthcare’s Enterprise Imaging ...
The American Society for Radiation Oncology (ASTRO) issued an updated clinical practice statement for accelerated partial breast irradiation (APBI) for early-stage breast cancer. The updated guideline reflects recent evidence that greater numbers of patients can benefit from accelerated treatment, and also provides direction for the use of intraoperative radiation therapy (IORT) for partial breast irradiation.
Data are streaming faster and in greater quantities than ever before. These data take the form of pictures coming from imaging machines. They may be ECGs, digital slides from pathology, or vital signs captured by wrist-wrapped fitness devices.
Despite decades of progress in breast imaging, one challenge continues to test even the most skilled radiologists ...
Eizo Inc. announced the release of the RadiForce RX660, a 30-inch, 6-megapixel monitor ideal for multimodality applications. It is the successor model to the RadiForce RX650 and features new functionality for improving reading room efficiency.
Carestream Health will showcase its MyVue Center Self-Service Kiosk, designed to enhance patients’ ability to access and manage their medical exam records, at the 2016 Radiological Society of North America conference, Nov. 27-Dec. 1 in Chicago. When this product is available, patients can use the kiosk to print or share their radiology reports and output their medical images onto radiographic film or a USB device.
Dilon Diagnostics and GE Healthcare announced the signing of an agreement for Dilon to be the exclusive distributor of GE's Discovery NM750b molecular breast imaging system in North America. Last year, GE Healthcare and Dilon Diagnostics signed a global distribution agreement in select world markets.
Bayer Radiology’s Barbara Ruhland and Thom Kinst discuss how radiology departments can address the many different ...
November 16, 2016 — JPI Healthcare announced the release of X-Vue DR Solutions, a family of digital radiography (DR) ...
Double Black Imaging’s 4K LED displays are designed to meet the demanding requirements for clinical review, modality imaging, surgical suites, operating rooms, emergency rooms and procedural facilities. These 8 mega-pixel displays (available in 27-, 31- and 55-inch models) provide superior imaging solutions with quadruple the resolution of most surgical displays available currently, according to the company.
King Edward VII’s Hospital in London, U.K., announced a new partnership with advanced imaging and informatics company Vital Images Inc. (Vital), based in Minneapolis, Minn., U.S.A.
The purpose of this exploratory video roundtable is to examine the state of breast biopsies today and to communicate the current challenges and inefficiencies affecting personnel that perform these important procedures. The video roundtable posed key questions to Key Opinion Leaders from the Cleveland Clinic on obstacles they face when performing stereotactic-guided breast biopsies, and why it is necessary to consider the ramifications. The roundtable discussion was hosted by Imaging Technology News and was funded through a grant from Hologic Inc.
TeraRecon, together with full-color 3-D printing cloud provider WhiteClouds, announced new technological and workflow process to print highly detailed models directly from the TeraRecon software. The new 3D Print Packs make online, cloud-based 3-D reconstruction and printing simple, fast and affordable.
November 15, 2016 — Spellman High Voltage Electronics Corp. announced their DC high-voltage power supplies, X-ray ...
Sorna Corp. announced that it has received certification from British Standards Institution Group America Inc. that Sorna complies with the requirements of the internationally recognized ISO 13485:2003 standard. The standard is applied for the Quality Management Systems of organizations involved in the design, development, testing, manufacture and distribution of medical devices.
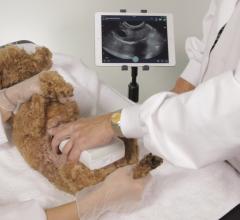
Clarius Mobile Health has introduced a new ultrasound scanner designed for veterinarians who treat household pets. The Clarius C7 Wireless Ultrasound Scanner pairs with iOS and Android devices to enable quick scans.

 November 18, 2016
November 18, 2016