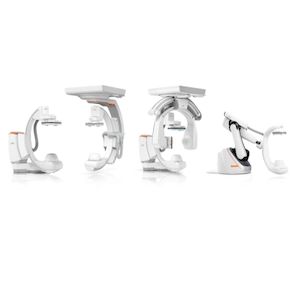
Artis interventional imaging portfolio. Photo: Siemens Healthineers
May 12, 2026 — Siemens Healthineers has received clearance from the Food and Drug Administration for six new systems in its Artis interventional imaging portfolio, which features the new Optiq AI imaging chain powered by artificial intelligence.
The new interventional systems include the floor, biplane, ceiling and pheno (or floor-mounted robotic) versions of the Artis vision platform; as well as the floor configurations of the Artis icono.explore and Artis genio platforms. The Optiq AI imaging chain common to all six systems uses AI to analyze and optimize image data in real time.
The six new systems address a wide range of clinical uses, operational needs, and financial considerations. The four systems in the Artis vision platform deliver high speed and precision for image‑guided procedures. The floor version of the Artis icono.explore is engineered for high patient throughput, and the Artis genio floor is a workhorse system designed for versatility and high productivity to handle a broad case mix. The unifying foundation across these six systems is the Optiq AI imaging chain, which meets the growing need for precision and premium image quality in image-guided procedures. Optiq AI applies deep‑learning‑based noise reduction across 2D imaging modes such as fluoroscopy, acquisition, and digital subtraction angiography. This denoising enables consistent, high-quality visualization during interventional procedures.
“As more complex procedures come to interventional labs, increased precision is of paramount importance,” said Kris McVey, head of Angiography in the Advanced Therapies business at Siemens Healthineers North America. “Our new Artis portfolio powered by Optiq AI delivers the high level of image quality clinicians rely upon to guide these intricate procedures—while taking into account the needs and budget of every type of healthcare facility.”
Additional Featues
Additional features in the Artis portfolio include advanced capabilities across procedures and a standardized user experience. Advanced 3D applications such as syngo DynaCT MORE reduce motion-related artifacts to enhance intraprocedural 3D imaging. The Touch UI touch screen enables tableside access to key system functions and supports direct, in‑case interaction as well as physician/technologist collaboration. Consistent controls across the portfolio help teams move between rooms with minimal variability.
To learn more, visit www.siemens-healthineers.com/en-us/angio/lp/artisicono


 May 11, 2026
May 11, 2026 









