March 23, 2026 —Sacumen has launched ConnectX, a unified AI platform that gives cybersecurity product companies full command of their connector ecosystems — from initial deployment and validation to continuous testing, real-time monitoring, and around-the-clock support. The platform made its global debut at RSA Conference (RSAC) 2026 in San Francisco.
Today's cybersecurity platforms run on integrations — SIEM, SOAR, XDR, IAM, and dozens more. But keeping those connectors current is a relentless challenge. Frequent API changes, shifting schemas, and sprawling tech ecosystems consume engineering bandwidth that should be driving product innovation — while the average enterprise loses over $1.2M a year to integration breakdowns. ConnectX was built to solve that problem.
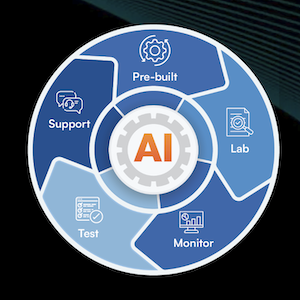
Five Pillars of Connector Excellence
- Prebuilt — 1,050+ production-ready connectors with full source code ownership across SIEM, SOAR, XDR, EDR, DSPM, IAM, MDR, and 65+ categories. With 82+ new connectors added monthly, organizations cut integration build time from months to weeks and expand their ecosystems 80% faster.
- Lab — Production-grade validation spanning 520+ live and virtual OEM environments — not dev mocks or static stubs. ConnectX Lab catches integration failures before they reach production, cutting UAT cycles by 50% and reducing rollback risk by 60%.
- Monitor — Agentic AI monitoring that tracks API drift across source-side OEM platforms and destination systems — not just the connectors Sacumen built. ConnectX surfaces schema and version changes before they impact production, alerting engineering teams before customers ever see a problem.
- Test — Automated AI agent testing across six validation dimensions — Configuration, Data Ingestion, Data Loss, Data Model Mapping, Action Execution, and Load Testing — with issues surfaced in under four hours. Each cycle delivers an executive health summary and a developer-level failure log.
- Support — SLA-backed, round-the-clock L2 and L3 support from engineers with deep connector expertise. ConnectX cuts break-fix resolution time by 65%, closing most issues within 18 hours and delivering change impact assessments within five business days.
Leadership Perspectives
"Connectors are no longer simple integrations — they are long-lived product infrastructure. Yet the industry has treated them as a one-time build problem. ConnectX changes that entirely. We're giving cybersecurity companies a single platform to own their connector lifecycle end-to-end, with AI at the core," said Nitesh Sinha, founder & CEO, Sacumen
Praneeth Kudithipudi, VP, Sacumen, said "The market has been waiting for a solution that goes beyond connector delivery and actually keeps them running reliably. ConnectX does exactly that — and the response from prospects has been extraordinary."
Learn more at www.sacumen.com.


 May 20, 2026
May 20, 2026 









