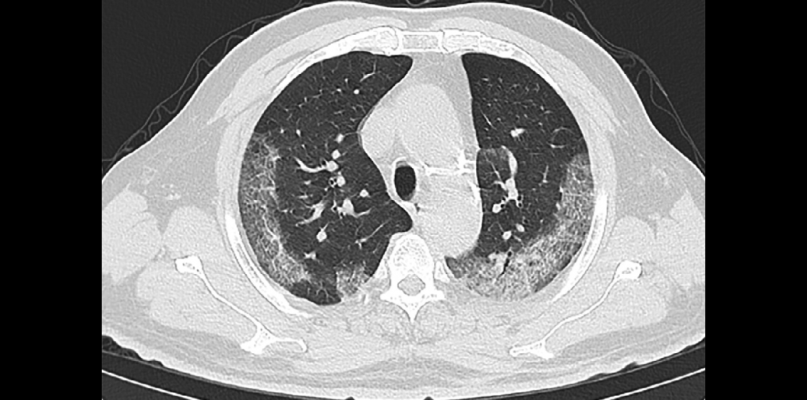
Examples of typical chest CT findings compatible with COVID-19 pneumonia in patients with epidemiological and clinical presentation suspicious for COVID-19 infection. This image is part of the original research, Sensitivity of Chest CT for COVID-19: Comparison to RT-PCR, published Feb. 19, 2020, in Radiology Online.
February 19, 2020 — In new research published in the journal Radiology, researchers in China cite the need for computed tomography (CT) in diagnosing patients with suspected novel coronavirus COVID-19, particularly when the lab test is negative. This is the first paper to closely examine the performance of CT vs. viral nucleic acid detection using real-time polymerase chain reaction (RT-PCR) for COVID-19 infection.
Although RT-PCR has been the standard of reference in diagnosing COVID-19, in this series of 51 patients with chest CT and RT-PCR assay performed within 3 days, the sensitivity of CT for COVID-19 infection was 98% compared to RT-PCR sensitivity of 71 percent.
The purpose of this study was to compare the sensitivity of chest CT and viral nucleic acid assay at initial patient presentation.
The findings suggest that if patients are only tested with RT-PCR, and the result is negative, they could be released to spread the infection.
“In our series, the sensitivity of chest CT was greater than that of RT-PCR. Our results support the use of chest CT for screening for COVD-19 for patients with clinical and epidemiologic features compatible with COVID-19 infection particularly when RT-PCR testing is negative,” the authors wrote.
You can read the full study, Sensitivity of Chest CT for COVID-19: Comparison to RT-PCR, here.
Additional References on the Novel Coronavirus:
1. Chest CT for Typical 2019-nCoV Pneumonia: Relationship to Negative RT-PCR Testing: Radiology. https://doi.org/10.1148/radiol.2020200343. Accessed Feb. 14, 2020
2. Time Course of Lung Changes On Chest CT During Recovery From 2019 Novel Coronavirus (COVID-19) Pneumonia: Radiology. https://doi.org/10.1148/radiol.2020200370. Accessed Feb. 14, 2020
3. U.S. Food and Drug Administration: https://www.fda.gov/news-events/press-announcements/fda-takes-significant-step-coronavirus-response-efforts-issues-emergency-use-authorization-first?utm_campaign=020420_PR_FDA%20Issues%20EUA%20for%20First%202019%20Novel%20Coronavirus%20Diagnostic&utm_medium=email&utm_source=Eloqua. Accessed Feb. 14, 2020.
4. Public Health News Alert: CMS Develops New Code for Coronavirus Lab Test. https://www.cms.gov/. Accessed Feb. 14, 2020.
Related Coronavirus Imaging Content:
Radiologists Describe Coronavirus CT Imaging Features
CT Imaging of the 2019 Novel Coronavirus (2019-nCoV) Pneumonia
Infervision in the Frontlines Against the Coronavirus
CT Imaging Features of 2019 Novel Coronavirus (2019-nCoV)
Chest CT Findings of Patients Infected With Novel Coronavirus 2019-nCoV Pneumonia


 May 26, 2026
May 26, 2026 









