April 5, 2018 — Medical imaging company Brainomix has attracted £7m ($9.8 million) of investment to bring its artificial intelligence (AI) software for fast stroke diagnosis and treatment to the global healthcare market.
Boehringer Ingelheim Venture Fund (BIVF), the corporate venture fund of the global pharmaceutical company Boehringer Ingelheim, joined the round, which was led by university investment veterans Parkwalk Advisors. Existing investors Chimera Partners and Oxford University Innovation Fund also participated.
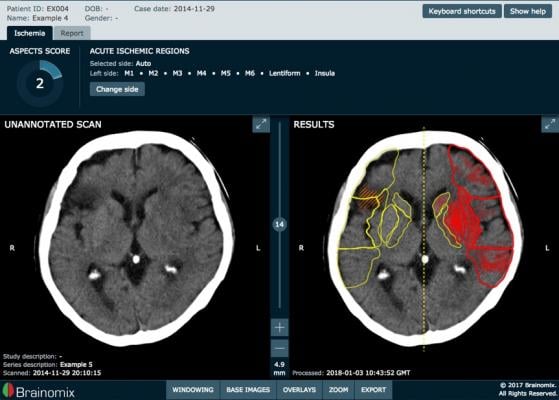
Brainomix's flagship software solution, e-ASPECTS, uses AI to provide a fast and standardized assessment of stroke patient computed tomography (CT) scans, supporting quick and consistent treatment decisions by medical professionals, irrespective of their experience and expertise. The software works by automating the clinically validated Alberta Stroke Program Early CT Score (ASPECTS) method. e-ASPECTS provides physicians with a clinical support tool that is designed to improve stroke treatment outcomes at much lower overall cost.
The injection of cash will allow Brainomix to further commercialize its intelligent medical imaging and workflow software solutions and expand its market penetration around the world.
For more information: www.brainomix.com


 May 19, 2026
May 19, 2026 









