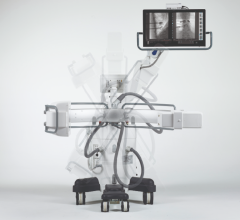
May 4, 2012 — Ziehm Imaging has been involved in mobile X-ray-based imaging for 40 years and is a major player in mobile C-arms. Since the company was founded in 1972, more than 10,000 Ziehm imaging C-arms have been installed in hospitals and medical facilities around the globe.
The Nuremberg-based company, with offices in the United States, Italy, France, Finland, Russia, Brazil, China, Singapore and South Africa, specializes in the development and production of mobile C-arms. Its range of products targets clinical users' needs, e.g., portable devices for emergency medicine in mobile operating room (OR) units and liquid-cooled high-end devices for hybrid operating rooms.
"Our strength lies in our dedicated focus on intraoperative imaging solutions in combination with our global distribution network covering more than 75 countries," says Martin Törnvik, vice president global sales and marketing of Ziehm Imaging GmbH.
As part of its global "40 Years of Innovation" campaign, Ziehm Imaging is seeking to convey its mobile C-arm expertise to its target groups. The C-arm specialist invests around 15 percent of its annual turnover in research and development. In the past 40 years, Ziehm Imaging has brought numerous new developments in the area of mobile X-ray technology onto the market and is the market leader in numerous countries. Just last year, Ziehm Imaging presented the hybrid edition of the Ziehm Vision RFD, a cost-effective, high-performance alternative to fixed installed systems in hybrid rooms.
With a product portfolio in which 80 percent of devices are less than three years old and with exciting innovations in development, the company is in an ideal position to further increase its share of the global market in the years to come.
For more information: www.ziehm.com


 April 15, 2026
April 15, 2026