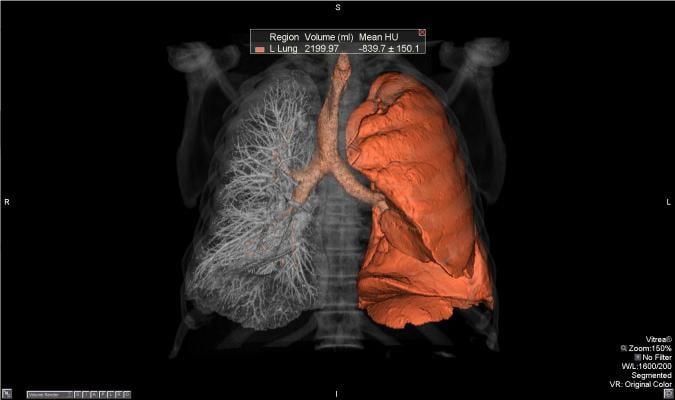
September 13, 2016 — Vital Images Inc. recently announced the support of Nuance PowerScribe 360 Reporting and PenRad PenLung Lung Screening Management system in its comprehensive Lung Screening solution. The Vitrea CT Lung Analysis application helps clinicians standardize the diagnosis of lung nodules, while the solution’s workflow system facilitates management of lung screening treatment plans.
Nuance’s PowerScribe 360 is a speech recognition technology for radiology, providing accurate, flexible and rapid reporting that is consistent and actionable. The high-quality reports generated by PowerScribe 360 can help healthcare organizations improve both patient care and physician satisfaction, and optimize billing and reimbursement.
PenLung by PenRad is a unified software solution that helps radiology departments and imaging facilities manage patients participating in low-dose computed tomography (LDCT) lung screening programs. It collects smoking and environmental risk history through automated web and tablet calculators to manage patient eligibility and export of the lung registry for reimbursement.
For more information: www.vitalimages.com


 May 19, 2026
May 19, 2026 









