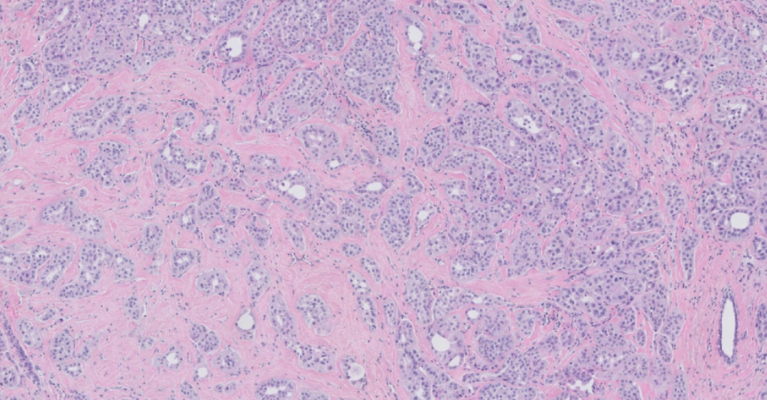
April 9, 2024 —PreciseDx, a leading innovator in oncology diagnostics leveraging Artificial Intelligence (AI) for revolutionary, morphology-driven disease analysis, announced a sponsored research collaboration with UCLA's Department of Pathology and Laboratory Medicine in Los Angeles to evaluate PreciseBreast's ability to accurately assess risk of recurrence for patients with triple-negative breast cancer (TNBC). TNBC constitutes only 10-20% of all breast cancer cases, yet continues to demand significant clinical interest due to its limited response to conventional treatments, the association with younger women, BRCA1/2 mutations, ethnic/racial disparities, and its highly aggressive characteristics.
The current study will expand on the previous subgroup analysis of TNBC patients from the PreciseBreast-Mount Sinai Hospital, NY, validation study with a large, well-annotated outcome-based TNBC cohort from the UCLA Health System.
TNBC represents a complex group of tumors that are defined by their absence of hormone receptors and the amplification of the HER2 gene, which has made effective treatments challenging. Recent developments including the existence of subtypes within the category of TNBC along with the role of immunotherapy have emphasized the need for more advanced tools to improve TNBC categorization. Early risk assessment is crucial for the effective management and prognosis of TNBC patients, as it enables timely intervention and more personalized treatment approaches.
Commenting on the significance of the collaboration, Dr. Nicholas Stanzione, Assistant Clinical Professor of Pathology and Laboratory Medicine, stated, "Risk assessment regarding the likelihood of disease recurrence is a critical first step in the phenotypic characterization of all cancer diagnoses, especially in aggressive subtypes like triple-negative breast cancer, which is often associated with poor patient outcomes because of the development of metastases in secondary organs including the brain, bone, and lungs. Our collaboration with PreciseDx has the potential to greatly improve our approach to predicting and managing recurrence in TNBC patients, ultimately leading to enhanced outcomes."
Integrating advanced AI algorithms and comprehensive clinical data, the collaborative study aims to confirm PreciseBreast as a highly effective and reliable digital breast cancer assay. This new assay will potentially enhance the early detection of TNBC recurrence and support personalized treatment strategies tailored to individual patient needs.
"We are excited to partner with UCLA Health in this innovative study," said Michael J. Donovan, PhD, MD, Co-Founder and Chief Medical Officer of PreciseDx. "By combining UCLA's clinical expertise and curated TNBC population with PreciseDx's cutting-edge AI technology, we aim to support the current standard of care by providing an AI-phenotype of TNBC to ultimately guide effective treatment decisions."
The collaborative study between UCLA and PreciseDx represents a significant step forward in digital pathology and oncology breast cancer research. It underscores the commitment of both institutions to advancing precision medicine and improving patient care.
For more information: www.precisedx.ai


 May 19, 2026
May 19, 2026 









