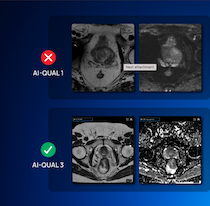
June 26, 2025 – Quibim, a global provider of quantitative medical imaging solutions, has launched AI-QUAL, a new feature to Quibim’s QP-Prostate platform, that automates the assessment of prostate MRI image quality in accordance with the PI-QUAL v2 guidelines. This marks a significant step forward in standardizing prostate MRI quality, enhancing diagnostic confidence, and streamlining radiology workflows.
AI-QUAL leverages advanced artificial intelligence to evaluate prostate MRI studies and assign a standardized score from 1 to 3, reflecting the diagnostic quality of the images. The system automatically detects common artifacts — such as rectal gas distortion and metal interference—that can compromise image quality and reduce diagnostic accuracy.
Recent studies have shown that one in three prostate MRI scans in the U.S. are not of diagnostic quality, underscoring the urgent need for automated, objective quality control tools in prostate imaging.
Key Benefits of AI-QUAL include:
- Automated Quality Scoring: Eliminates manual checks, saving time and reducing subjectivity
- Standardized Reporting: Delivers consistent quality score based on PI-QUAL v2 guidelines
- Artifact Detection: Identifies key image-degrading factors such as rectal gas distortion and metal artifacts
- Diagnostic Confidence: Ensures radiologists work with the highest quality images
- Seamless Integration: Designed to fit effortlessly into existing radiology workflows
David Bazaga, VP of Product at Quibim, commented, “AI-QUAL represents a major advancement in our mission to transform imaging into a driver of precision health. By automating image quality assessment, we’re helping radiologists reduce variability, improve diagnostic accuracy, and ultimately deliver better outcomes for patients.”
Low-quality imaging has been linked to an increase in PI-RADS 3 findings, which can lead to unnecessary biopsies and negatively impact patient care. AI-QUAL addresses this challenge by providing a standardized, automated approach to image quality assessment, helping clinicians make more confident and informed decisions.
“We are proud to introduce AI-QUAL as part of our growing suite of prostate imaging solutions. This tool directly addresses a critical gap in prostate MRI quality control and reinforces our commitment to supporting radiologists with cutting-edge, clinically validated AI technologies,” said Dr. Ángel Alberich-Bayarri, Quibim CEO and founder.
AI-QUAL is now available as part of Quibim’s QP-Prostate platform, further strengthening its position as a comprehensive solution for prostate cancer detection, diagnosis, and care.
To learn more about Quibim visit https://quibim.com/.


 May 12, 2026
May 12, 2026 









