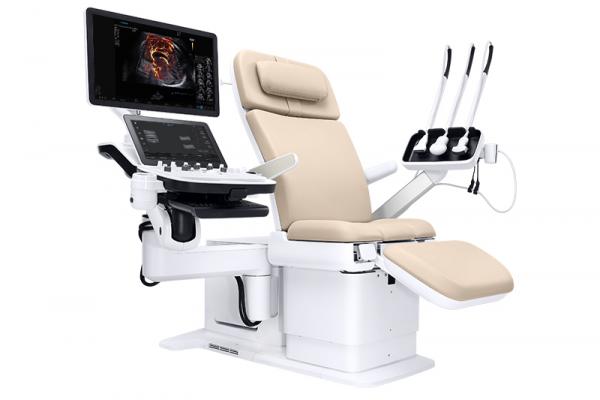
September 27, 2019 — NeuroLogica, a subsidiary of Samsung Electronics Co. Ltd., announced that the Samsung Hera I10 ultrasound system has received U.S. Food and Drug Administration (FDA) clearance and will be unveiled at the Society of Diagnostic Medical Sonography (SDMS) 2019 Annual Conference, Sept. 26-29 in National Harbor, Md. The Hera I10 is the latest addition to the Samsung Hera family of Premium Women’s Health ultrasound systems and represents a first of its kind in ultrasound system design, according to Samsung, with the integration of an ultrasound examination chair.
Through its integrated ultrasound exam chair, ergonomics and technology, the Hera I10 aims to improve the ultrasound experience, clinical confidence and ultimately, patient care. The system is currently available commercially in the U.S.
The design of the Hera I10 aims to increase comfort and safety, resulting in a smoother and more relaxing ultrasound exam for both the clinician and patient.
-
Integrated, Motorized Chair: Enables clinicians to help patients safely and comfortably move into the optimal position to capture the images needed to provide a confident diagnosis;
-
Smooth Adjustability for Patient Comfort: Users can adjust the chair from 18.9 inches in the start position to a maximum height of 38.6 inches, eliminating the need for patients to maneuver uncomfortably onto a traditional exam table;
-
Durability for Long-Lasting Use: The exam chair is designed to withstand up to 440 pounds, enabling it to withstand long-term use.
Hera I10’s new design has an extremely wide range of motion, allowing the system to adapt to the unique scanning position of each user and creating an optimized scanning environment to help reduce pain and strain while scanning.
-
Floating Console: Allows technicians to position the system where they need it, enabling all-day scanning comfort. Internal studies have shown an increase in user satisfaction of 50 percent, and a reduction in the force required to move the system back and forth between abdominal and vaginal scanning by approximately 52 percent when compared to traditional ultrasound.
-
Transducer Station and Cable Support Arm: Supports the majority of the transducer cable’s weight to reduce force on the scanning wrist by 70 percent, as found by the aforementioned internal study;
-
Motorized, Adjustable Exam Chair: Helps clinicians safely assist patients on and off of the exam table as well as into a reclined position for their ultrasound exam, helping reduce associated risk of injury while performing this common task.
As the newest addition to the Hera platform, the Hera I10 incorporates Samsung’s Crystal Architecture to enable image clarity and advanced technology for confident diagnosis.
-
ShadowHDR: This proprietary technology helps suppress shadows caused by fetal limbs or bone to reveal details otherwise obscured;
-
LumiFlow: Provides dimensional visualization of blood flow that aids in quickly understanding vessel boundaries, and may provide additional spatial comprehension when documenting fetal and maternal hemodynamics; and
-
MV-Flow: Provides detailed visualization of microvascular perfusion into tissues and organs, helping to easily visualize fetal lung perfusion, ductus venosus, MCA and adnexal low-flow hemodynamics.
“As a maternal fetal specialist who receives the most challenging OBGYN referral cases, I feel like it’s my responsibility to have the best equipment in town, which is exactly what Hera I10 provides,” said Janet Davis, M.D., F.A.C.O.G. “With the growing obesity epidemic, it is especially important to have an ultrasound machine that provides the highest imaging quality in order to reveal fetal detail that we previously could not see. I also love the Hera I10’s ergonomic design, which increases comfort for sonographers while scanning and can lead to increased productivity and less on-the-job strain.”
For more information: www.samsunghealthcare.com


 May 07, 2026
May 07, 2026 









