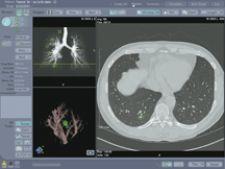
With R2’s Version 2.1 ImageChecker CT Lung CAD System, results from the AutoPoint Temporal Comparison tool (for tracking lung nodule growth between current and prior multislice CT exams) can now be sent to a PACS workstation for full enterprise accessibility, without the need for R2’s dedicated CT CAD workstation. According to the manufacturer, with the new Version 2.1 software, R2’s Pulmonary Artery Patency Exam (PE) tool features a new algorithm to aid physicians in more effectively detecting intravascular filling defects, such as pulmonary embolism, during review of multislice CT exams.


 June 06, 2023
June 06, 2023 









