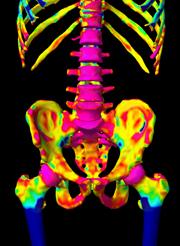
3-D thickness map
November 15, 2010 — A new method for identifying which bones have a high risk of fracture and for monitoring the effectiveness of new bone-strengthening drugs and techniques has been developed by scientists at the University of Cambridge in Cambridge, UK. The method, developed by Dr. Graham Treece of the Department of Engineering and Dr. Ken Poole of the Department of Medicine, uses computed tomography (CT) imaging to accurately measure the thickness of the cortical bone, the hard outer layer of compact bone which surrounds the spongy trabecular bone. The thickness of cortical bone is a key indicator of the risk of fracture, including in conditions such as osteoporosis.
When CT rays pass through thin, radiodense layers such as the cortical bone, the resulting images are slightly blurred. Understanding the mathematics of how this blurring occurs has allowed the researchers to develop an image analysis process that provides an accurate measurement of the cortical thickness at a particular point on the bone. Importantly, this method remains accurate in the clinically relevant sub-millimeter range and can be achieved using routine CT scans.
The cortical thickness measurement can be obtained for thousands of points on the surface of a bone, thus providing a thickness map. This, in turn, allows clinicians to identify bones that exhibit thinning of the cortical bone at the key points that are known to be associated with an increased risk of fracture. Successive mapping over time allows maps to be produced depicting changes in the cortical bone over time in all areas of the skeleton, so that the progress of osteoporosis treatments can be monitored.
The current technologies do not provide for this level of functionality. Dual-energy X-ray absorptiometry (DXA) provides a measure of mineral levels across the whole bone, not a 3-D map, whereas multi-detector computed tomography (MDCT) does not achieve the sub-millimeter resolution provided by this novel approach.
“Osteoporosis is a condition which will become more and more common as the population ages,” said Dr. Andrew Walsh of Cambridge Enterprise, the University’s commercialization group. “This exciting innovation has the potential to improve the treatment of patients suffering from osteoporosis, as well as to support the development of new drugs. We are currently seeking commercial partners to help us achieve this benefit by bringing this technology to market.”
This work will be demonstrated at RSNA 2010.
For further information: www.enterprise.cam.ac.uk


 May 04, 2026
May 04, 2026 









