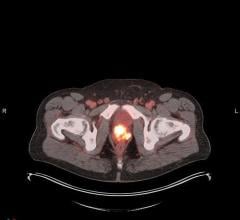
June 17, 2009 – Molecular Insight Pharmaceuticals Inc. presented clinical data on a radiolabeled, small-molecule molecular imaging pharmaceutical in development for diagnosis and staging of prostate cancer at the 2009 annual meeting of the Society of Nuclear Medicine (SNM) in Toronto.
Trofex, which targets prostate specific membrane antigen (PSMA), a protein highly expressed by prostate tumor cells, has the potential to both detect and, when labeled with a therapeutic isotope, to treat metastatic prostate cancer.
In 2008, Molecular Insight initiated clinical studies of Trofex in patients with documented prostate cancer and confirmed hormone refractory metastatic disease. Two internally developed Trofex compounds, MIP-1072 and MIP-1095, were evaluated for their capacity to visualize the disease and to define their pharmacokinetic profiles. Both agents rapidly detected metastatic prostate cancer lesions in soft tissues and bone, confirming that targeting the extracellular domain of PSMA is a viable approach for the molecular targeting of this cancer. The compounds were uniquely able to detect metastases within one to two hours after injection.
As John W. Babich, Ph.D., Executive Vice President, Chief Scientific Officer and President of Research and Development at Molecular Insight Pharmaceuticals, indicated, “Not only does this data demonstrate the capability to rapidly visualize bone metastasis and soft tissue lesions — that may be missed by conventional imaging techniques — imaging can be done in a single day, which is not possible with some of the products currently on the market for imaging recurrent metastatic disease.
Abstract Title:
Targeting Metastatic Prostate Cancer (PCa) in Patients using 123I-MIP 1072 & 123I-MIP 1095
Authors:
J.A. Barrett1; N.D. LaFrance1; R.E. Coleman2; S.J. Goldsmith3; J.B. Stubbs4; N. Petry2; S. Vallabhajosula3; K.P. Maresca1; F. Femia1; M. Stabin4; J.W. Babich1
1Molecular Insight Pharmaceuticals, Inc., Cambridge, MA, 2Duke University, Durham, NC ,3Cornell Medical Center, NY, NY 4, Radiation Dosimetry Systems, Inc, Palo Alto, CA
For more information: www.molecularinsight.com


 May 06, 2026
May 06, 2026