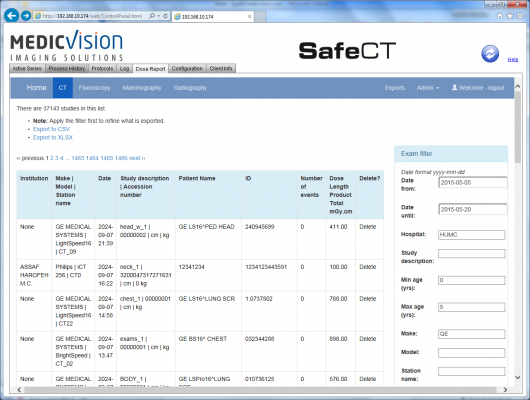
May 27, 2015 — Medic Vision Imaging Solutions Ltd. announced the introduction and availability of SafeCT Dose Reporting. Healthcare facilities can now, for the first time, use a single software platform to meet their low dose computed tomography (CT) imaging needs while also conforming to CT dose monitoring and reporting standards.
SafeCT Dose Reporting is an extension of the SafeCT iterative image reconstruction product line. It continuously captures the dose information directly from the CT scanner and saves it to a database for easy retrieval and analysis. Reports can be generated from the database as needed — for example, per patient, per day, per scanner or per procedure. The solution was designed to help healthcare facilities be compliant with The Joint Commission dose reporting requirements that will become effective July 1, 2015.
SafeCT Dose Reporting is currently available and has already been implemented by healthcare facilities in the United States, including Yakima Valley Memorial Hospital (YVMH), Yakima, Washington. YVMH, which conducts approximately 5,000 CT exams a year, has been offering low-dose CT scans using SafeCT since 2013. The facility is now using the new Dose Reporting function with its three CT scanners to benefit from dose reduction, monitoring and reporting.
Available to both existing and new Medic Vision customers, SafeCT Dose Reporting is an enhancement to the existing SafeCT product. It can be used in conjunction with the Safe CT low-dose CT image enhancement solution, or as a stand-alone dose reporting system. Vendor neutral, the solution works with all CT scanners, of all vendors and models.
For more information: www.medicvision.com


 April 29, 2026
April 29, 2026 









