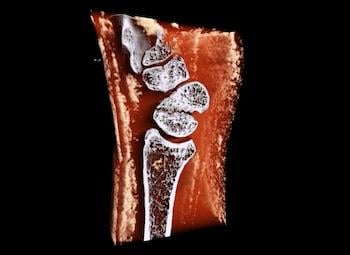
MARS scan of a wrist with a scaphoid fracture
Aug. 12, 2025 — MARS Bioimaging has appointed of Dr. Ojas Mahapatra as Group Chief Executive Officer. Mahapatra is an accomplished executive with more than a decade of experience scaling and transforming deep-tech companies in global markets. Mahapatra most recently was CEO of New Zealand cleantech company Fabrum.

The New Zealand-headquartered medical device company specializes in spectral molecular imaging CT scanners that deliver 3D color X-rays for point-of-care diagnosis.
“We’re thrilled to welcome Dr Mahapatra as Group CEO to lead MARS in the exciting clinical-pathway phase, where his efforts will be focused on placing growth capital and talent to accelerate growth," said Christopher Stoelhorst, Chairman of MARS Bioimaging, "His leadership will be instrumental in scaling our commercial and go-to-market capability, advancing entry into new markets internationally where the deployment of the MARS diagnostic product will improve health economics and health equity.”
The company also announced it is advancing the final phase of its Series A investment round to support clinical commercialization milestones. Following a successful initial close of $7.6 million last year, the company is now selectively engaging with new investors to support its next stage of scale, with a total raise target of $10 million under the same terms as the initial close.
For more information on the company, please visit www.marsbioimaging.com


 May 04, 2026
May 04, 2026 









