September 8, 2014 — iCAD Inc. announced that, in addition to Volpara Density volumetric breast density assessment software, Volpara Analytics and Volpara Dose solutions are now available as part of iCAD’s PowerLook Advanced Mammography Platform (AMP). With the addition of these quantitative breast-imaging tools, radiologists will be able to evaluate the performance of all major digital mammography and tomosynthesis units and obtain patient-specific dose information to make informed treatment decisions in real time.
“Today’s healthcare environment is increasingly data-driven and our customers require up-to-the-minute access to analytics that impact patient care,” said Ken Ferry, CEO of iCAD. “By incorporating additional breast imaging analytic and reporting solutions from Volpara, our PowerLook AMP customers will be able to go beyond automated mammography unit settings and mine patient-specific, real-time information for actionable insights regarding care delivery within their practices and across patient populations.”
PowerLook AMP is a next-generation digital mammography computer-aided detection (CAD) platform that provides radiologists with the ability to customize their 2-D CAD solution to meet the needs of their individual work environment. Now with the option to incorporate VolparaAnalytics and VolparaDose in addition to VolparaDensity, PowerLook AMP users will, among other benefits, be able to:
- Improve patient safety and reporting through personalized measurements of density, patient dose, breast compression and other volumetric factors designed to help maintain accuracy and consistent quality in breast screening.
- Optimize performance/utilization of staff and mammography systems.
- Utilize a globally-accepted algorithm incorporating breast density to generate patient-specific radiation dose estimates, which can be automatically entered in patient communications.
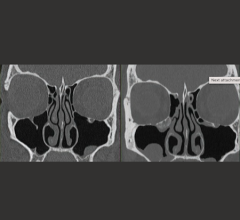
Cleared by the U.S. Food and Drug Administration (FDA), HealthCanada, the TGA and CE-marked, VolparaDensity is used by radiologists to objectively assess density from both digital mammography and tomosynthesis images to help doctors evaluate who might benefit from additional screening. Highly correlated to breast magnetic resonance imaging (MR) assessments, VolparaDensity is a reliable tool that automatically generates an objective measurement of volumetric breast density correlated to the ACR (American College of Radiology) breast density categories. To date, more than 3 million women have had their breast density analyzed using VolparaDensity.
For more information: www.icadmed.com


 April 20, 2026
April 20, 2026