April 21, 2011 – The U.S. Food and Drug Administration (FDA) has cleared Hologic to incorporate the U.S. National Health and Nutrition Examination Survey (NHANES) Whole Body Reference Data into its Discovery dual X-ray absorptiometry (DXA) systems. These body composition values are useful in cases when a disease or condition itself, or its treatment, can affect the relative amounts of fat and lean tissue.
The prevalence of obesity has reached pandemic proportions. Globally, 1.5 billion adults were overweight in 2008 and 65 percent of the world's population live in countries where the number of obese and overweight people now exceeds the underfed. According to the U.S. Centers for Disease Control and Prevention (CDC), approximately 72.5 million adults in the United States were considered obese in 2007-2008, increasing their risk for coronary heart disease, hypertension, stroke, type 2 diabetes, certain types of cancer and premature death. Unfortunately, children are not immune to this disease. It is estimated that 17 percent of American children and adolescents ages 2-19 years old are obese, according to the latest NHANES.
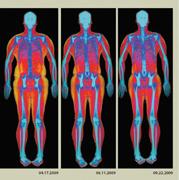
The FDA clearance of the Hologic Advanced Body Composition Assessment package opens up a new market for the Hologic Discovery whole body DXA systems that, up to now, have primarily been used to measure osteoporosis in women after they reach menopause.
"Body mass measurement on our Discovery systems is another significant milestone for Hologic and our skeletal health business," said John Jenkins, vice president and general manager of the Hologic Skeletal Health business. "Our new Advanced Body Composition assessment package provides a more detailed and more consistent baseline for measuring body mass changes than bio-impedance testing, hydrostatic weighing and skin fold thickness. This new indication is a major step forward in helping maintain the quality of life of men and women and, we hope, will become the standard of care for adults and children."
The assessment package is designed to provide a quick, accurate and precise low-dose X-ray exam to aid the physician in the assessment of fat, lean tissue and bone. Some of the diseases and conditions for which body composition values are useful include chronic renal failure, anorexia nervosa, obesity, AIDS/HlV, and cystic fibrosis. A whole body composition scan can take as little as three minutes. During the exam, the patient lies comfortably on a padded table while the system scans the body. Unlike typical X-ray machines, radiation exposure during this exam is extremely low and involves no injections, invasive procedures or pain. The new assessment package includes color images displaying the distribution of fat, lean tissue and bone and an easy to interpret report for improved patient management and counseling.
For more information: www.hologic.com


 May 07, 2026
May 07, 2026 









