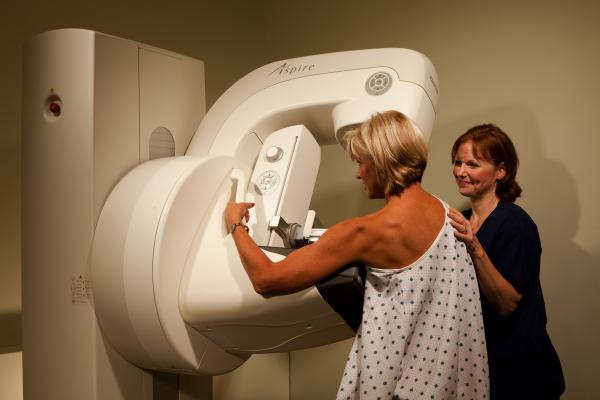
August 25, 2016 — Fujifilm Medical Systems U.S.A. has submitted to the U.S. Food and Drug Administration (FDA) the final module of its premarket approval (PMA) application for digital breast tomosynthesis (DBT), as an optional software upgrade for the Aspire Cristalle digital mammography system.
The optional DBT 3-D mammography upgrade for the Aspire Cristalle system, known as Amulet Innovality outside of the United States, has been available since May 2013 in Europe, Asia and Latin America.
“Today marks another milestone in Fujifilm’s mission to bring innovative digital breast tomosynthesis technology to physicians and women across America,” said Rob Fabrizio, director of strategic marketing, digital radiography and women’s health, Fujifilm Medical.
Aspire Cristalle is available in the United States today as a full field digital mammography (FFDM) system. Aspire Cristalle features Fujifilm's innovative hexagonal close pattern (HCP) detector pixel design, engineered for higher acquisition efficiency, to enhance detail for improved low dose performance compared to conventional square pixel design. The result is sharper images with gentler dose to the patient. Additionally, Aspire Cristalle incorporates a patient calming design and Fujifilm’s patented Comfort Paddle, which is designed to make the most physically unpleasant part of mammograms, more comfortable. The paddle’s unique soft edges, flexible composition and four-way pivot contours to the individual shape of the breast to more comfortably apply just the right compression for optimal tissue separation.
For more information: www.fujifilmhealthcare.com


 May 22, 2026
May 22, 2026 









