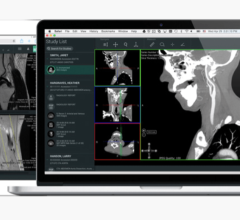
January 21, 2008 – The FDA cleared St. Jude Medical Inc.’s EnSite Fusion Registration Module, a software designed to help physicians create detailed heart models to facilitate the diagnosis and delivery of therapy for complex abnormal atrial heart rhythms, including Atrial Fibrillation (AF).
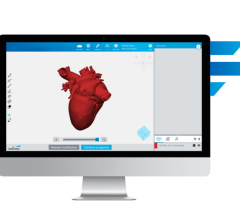
The software integrates an EnSite System-created chamber model with a 3D computed tomography (CT) model so that the physician has an improved view of the heart’s anatomy to better guide the delivery of therapy. EnSite Fusion also displays electrical information such as voltage, activation timing and lesion data directly on the CT model. In addition, it provides the flexibility to transition quickly between the fused model and the original EnSite model throughout the procedure, helping clinicians optimize the view to best meet the specific demands of the procedure.
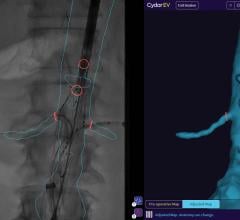
The EnSite System is used in minimally invasive electrophysiology procedures. Catheters with electrodes are inserted into the cardiac chamber and then are located or “sensed” by the EnSite System, which records electrical information and creates a rendering of the chamber anatomy.
For more information: www.sjm.com


 April 18, 2025
April 18, 2025