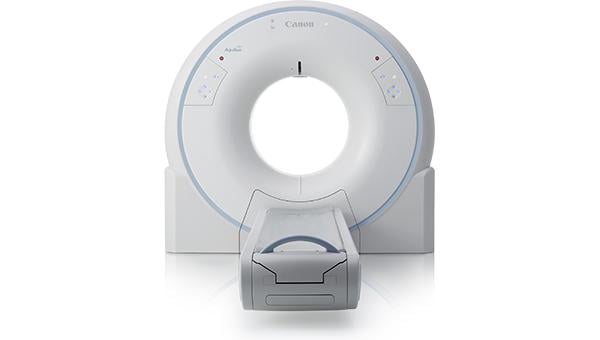
March 14, 2019 — Canon Medical Systems Europe B.V. introduced the all-new Aquilion Start computed tomography (CT) system to the European market at the 2019 European Congress of Radiology (ECR), Feb. 28-March 3 in Vienna, Austria. Featuring premium technologies based on Canon Medical’s Aquilion family, the new Aquilion Start will provide the opportunity for medical institutions to extend their radiology practice.
With the smallest installation footprint in its class, low power requirements and integrated dose-reduction technologies, Aquilion Start has the potential to fit into small X-ray rooms while providing high-quality 3-D CT imaging at lowest possible dose.
Built around the needs of patients and clinicians. Aquilion Start incorporates a spacious gantry to allow for scanning large patients in a comfortable environment. Intuitive controls and increased automation help clinical staff to quickly familiarize with system operation. Advanced technologies ensure optimized patient safety, accelerated clinical decision-making and improved workflow efficiency.
The system is developed and manufactured in Japan, and provides technologies also found in premium Canon CT systems:
- AIDR 3D Enhanced (Adaptive Iterative Dose Reduction) with up to 75 percent reduction in dose;
- PUREViSION Detector to optimize image quality and lower patient dose;
- SEMAR (Single Energy Metal Artifact Reduction) to facilitate confident diagnosis of patients with metal implants; and
- SURESubtraction for e.g. automated bone-free CT angiography of superior quality.
For more information: www.us.medical.canon


 May 19, 2026
May 19, 2026 









