March 28, 2022 — An artificial intelligence (AI) tool can accurately and consistently classify breast density on mammograms, according to a study in Radiology: Artificial Intelligence.
Breast density reflects the amount of fibroglandular tissue in the breast commonly seen on mammograms. High breast density is an independent breast cancer risk factor, and its masking effect of underlying lesions reduces the sensitivity of mammography. Consequently, many U.S. states have laws requiring that women with dense breasts be notified after a mammogram, so that they can choose to undergo supplementary tests to improve cancer detection.
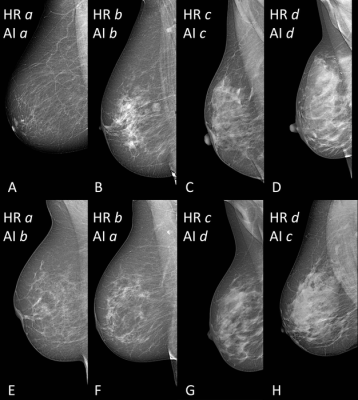
In clinical practice, breast density is visually assessed on two-view mammograms, most commonly with the American College of Radiology Breast Imaging-Reporting and Data System (BI-RADS) four-category scale, ranging from Category A for almost entirely fatty breasts to Category D for extremely dense. The system has limitations, as visual classification is prone to inter-observer variability, or the differences in assessments between two or more people, and intra-observer variability, or the differences that appear in repeated assessments by the same person.
To overcome this variability, researchers in Italy developed software for breast density classification based on a sophisticated type of AI called deep learning with convolutional neural networks, a sophisticated type of AI that is capable of discerning subtle patterns in images beyond the capabilities of the human eye. The researchers trained the software, known as TRACE4BDensity, under the supervision of seven experienced radiologists who independently visually assessed 760 mammographic images.
External validation of the tool was performed by the three radiologists closest to the consensus on a dataset of 384 mammographic images obtained from a different center.
TRACE4BDensity showed 89% accuracy in distinguishing between low density (BI-RADS categories A and B) and high density (BI-RADS categories C and D) breast tissue, with an agreement of 90% between the tool and the three readers. All disagreements were in adjacent BI-RADS categories.
“The particular value of this tool is the possibility to overcome the suboptimal reproducibility of visual human density classification that limits its practical usability,” said study co-author Sergio Papa, M.D., from the Centro Diagnostico Italiano in Milan, Italy. “To have a robust tool that proposes the density assignment in a standardized fashion may help a lot in decision-making.”
Such a tool would be particularly valuable, the researchers said, as breast cancer screening becomes more personalized, with density assessment accounting for one important factor in risk stratification.
“A tool such as TRACE4BDensity can help us advise women with dense breasts to have, after a negative mammogram, supplemental screening with ultrasound, MRI or contrast-enhanced mammography,” said study co-author Francesco Sardanelli, M.D., from the IRCCS Policlinico San Donato in San Donato, Italy.
The researchers plan additional studies to better understand the full capabilities of the software.
“We would like to further assess the AI tool TRACE4BDensity, particularly in countries where regulations on women density is not active, by evaluating the usefulness of such tool for radiologists and patients,” said study co-author Christian Salvatore, Ph.D., senior researcher, University School for Advanced Studies IUSS Pavia and co-founder and chief executive officer of DeepTrace Technologies.
For more information: www.rsna.org
Related Breast Density Content:
VIDEO: The Impact of Breast Density Technology and Legislation
VIDEO: Personalized Breast Screening and Breast Density
VIDEO: Breast Cancer Awareness - Highlights of the NCoBC 2016 Conference
Fake News: Having Dense Breast Tissue is No Big Deal
The Manic World of Social Media and Breast Cancer: Gratitude and Grief


 April 20, 2026
April 20, 2026 









