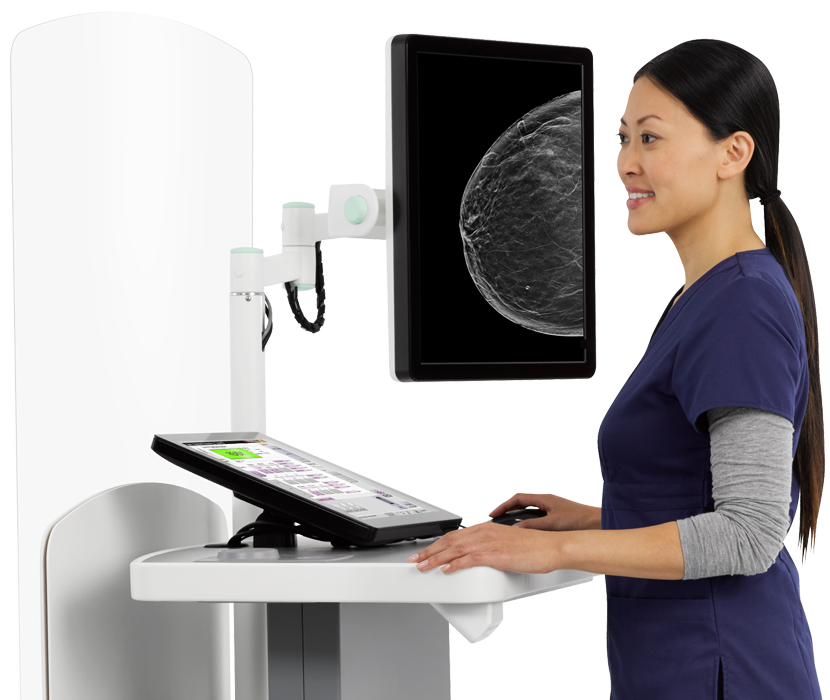
Photo courtesy of Hologic.
The global breast imaging market is expected to reach $7.3 billion by 2024, according to a new report by Grand View Research, Inc., a San Franciso-based market research and consulting company. The increasing prevalence of breast cancer and supportive government initiatives to increase awareness are expected to boost demand in the market.1
This market will primarily be driven by factors such as growing prevalence of breast cancer, increasing awareness about early detection of breast cancer and government investments, and funding for breast cancer treatment, said Research and Markets in its “Global Breast Imaging Market: Trends Analysis and Forecasts up to 2022” report. However, the growth is likely to be restrained by factors such as high installation costs of breast imaging systems, side effects of radiation exposure, and errors in breast cancer screening and diagnosis.2
Technological advances, such as molecular breast imaging, digital mammography, optical imaging, electrical impedance imaging and tomosynthesis are now propelling the market and are expected to show a lucrative growth over the forecast period. The innovation of complementary products to provide comfort to a woman undergoing screening is expected to catalyze the market growth further.1
Tomosynthesis
The 3-D tomosynthesis segment is identified as the fastest-growing segment of the breast imaging market. This technology offers enhanced efficiency and higher diagnostic accuracy in comparison to other available technologies, which is also expected to help boost market growth.1
At the recent 2018 Society for Breast Imaging (SBI)/American College of Radiology (ACR) Breast Imaging Symposium in Las Vegas, many current technologies were being showcased on the show floor. Hologic’s Clarity HD high-resolution 3-D imaging and Intelligent 2-D imaging technology recently received premarket approval (PMA) from the U.S. Food and Drug Administration (FDA) and are now available on the 3Dimensions breast tomosynthesis system. With these innovations, the system now provides higher resolution 3-D breast images for radiologists, enhanced workflow for technologists and a more comfortable mammography experience with low-dose options for patients.
Clarity HD high-resolution 3-D imaging technology provides fast, high-resolution 3-D images to accelerate screening and analysis, and is designed to clearly reveal subtle lesions and fine calcifications to help pinpoint cancers early. Clarity HD technology’s advanced detector and 3-D imaging algorithm work together to deliver high-quality 3-D images, regardless of breast size or density. Intelligent 2-D imaging technology works with Clarity HD technology to deliver high overall image quality, and clarity, contrast and detail at a lower dose.
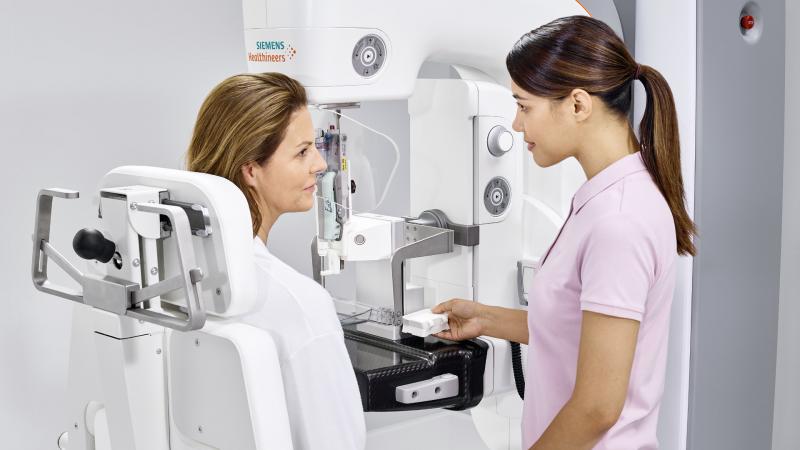
Siemens Healthineers’ HD breast tomosynthesis offers the widest image acquisition angle available at 50 degrees, according to the company. The wide angle delivers a high depth resolution for better separation of overlapping breast tissue, providing high-quality 3-D images for improved diagnostic confidence and earlier lesion detection. This technology is the basis for Mammomat Revelation’s HD Breast Biopsy solution, which enables one-click targeting of suspicious areas with a +/-1 mm accuracy.
Breast Density
Nearly half of all women in the United States who receive mammograms have dense breasts, complicating efforts to identify breast lesions. Typically, the radiologist assesses fibroglandular breast density visually after the patient has left the facility. The Siemens Mammomat Revelation offers automated breast density measurement during the mammogram for immediate, personalized risk stratification and more individualized care. Its soft compression adjusts the level of breast compression automatically to suit each patient’s anatomy. Coupled with ergonomic SoftComp Paddles, personalized soft compression enables improved breast positioning, more consistent image quality, and less discomfort and anxiety.
According to JoAnn Pushkin, DenseBreast-info, Inc.’s executive director, 34 states, encompassing more than 84 percent of American women, now have enacted laws that require some level of density notification following a mammogram. “Everyone needs to be aware, though, that not all of the laws, even if the state has a law that requires density notification, that information doesn’t necessarily provide women with information about her own breast density,” she stressed. “It might just be that the state is required to provide a general notification about breast density, so not all laws tell a woman she has dense breasts.”
The American College of Radiology (ACR) just released new recommendations on breast cancer screening in women with higher than average risk. “The fact that the ACR is recognizing who is at greater risk was a helpful clarification, and so as a breast cancer survivor, the two newest pieces that I think will affect screening protocols for women is that any woman who is diagnosed with breast cancer under the age of 50, no matter what her breast density is, are recommended to have an annual mammogram and MRI, and same thing for breast density and a history of breast cancer no matter what age she was diagnosed,” said Pushkin. “This will lead to earlier-stage diagnosis, which is what saves lives.”
Several software programs have been created to help radiologists better assess breast density. Densitas Inc. recently received FDA clearance for its Densitas|density automated breast density software. The software analyzes the same processed digital mammograms that radiologists view and are routinely stored in picture archiving and communication systems (PACS). The zero-click software provides radiologists with breast density assessments at point-of-care that are reportedly as accurate as the visual assessments made by experienced Mammography Quality Standards Act (MQSA)-qualified radiologists. The automated reports enhance confidence in follow-up care decisions and facilitate uniformity in density measurements across clinics and major hospital networks.
In addition, Hologic’s Quantra 2.2 Breast Density Assessment Software, also available on the 3Dimensions system, enables clinicians to provide women with consistent breast density assessments during routine breast cancer screenings. Through a proprietary algorithm powered by machine learning, Quantra software analyzes mammography images for distribution and texture of breast tissue, delivering clinicians accurate breast density assessment. The software categorizes breasts into four density categories, in alignment with the American College of Radiology Breast Imaging-Reporting and Data System (ACR BI-RADS) Atlas 5th Edition, according to the company.
Artificial Intelligence
Artificial intelligence (AI) is changing the world of radiology. In this month’s supplement, “How Artificial Intelligence Will Change Radiology,” ITN contributing editor Greg Freiherr takes a look at three key components of artificial intelligence. Rest assured AI will not be diagnosing patients and replacing doctors, he says — it will be augmenting their ability to find the key, relevant data they need to care for a patient and present it in a concise, easily understandable format.
Artificial intelligence definitely has its place in breast imaging as well. Ikonopedia introduced its next-generation breast ultrasound reporting module at the SBI/ACR Breast Imaging Symposium. The BI-RADS-compliant interface is intuitive, designed to streamline reporting for both screening and diagnostic exams acquired with both automated breast ultrasound (ABUS) and handheld ultrasound.
The new module is based on the teaching files of A. Thomas Stavros, M.D., FACR, one of Ikonopedia’s founders and author of the reference book “Breast Ultrasound” (Lippincott Williams and Wilkins, 2004). The module guides breast imagers through the interpretive process for breast ultrasound exams with an intuitive lesion locator tool designed specifically for breast ultrasound. It leverages Ikonopedia’s proprietary reporting interface and lesion locator that has increased efficiency for breast imagers in other breast imaging modalities. All reports are guaranteed to be 100 percent BI-RADS language-compliant and current with the Atlas 5th Edition. Ikonopedia is fast without sacrificing accuracy while eliminating reporting errors common with dictation, such as inappropriate BI-RADS, incomplete reports and wrong-side reporting, according to the company. Stavros said the module’s situational-based display of only relative icons unclutters the screen, and makes reporting cleaner, faster and more efficient.
Technologists are able to monitor their own performance and self-train with VolparaEnterprise software, which features a technologist dashboard. The lead technologist sees a Quality Quadrant diagram that summarizes patient positioning and compression performance by each technologist, helping to identify training opportunities. “We have been using VolparaEnterprise to redefine how to track and trend our technologist’s quality and facility workflow. VolparaEnterprise has provided our institute with amazing objective, analytic capability that has allowed us to applaud the team’s strengths and use them to facilitate collaborative growth, while identifying opportunities that we individualize by technologist. We have seen continued and sustained improvements as a result of this program,” said Maureen Mann, executive director of Boca Raton Regional Hospital’s Christine E. Lynn Women’s Health & Wellness Institute in Florida.
Utilizing state-of-the-art image analysis and deep learning technology, ScreenPoint’s Transpara automatically identifies soft-tissue and calcification lesions and combines the findings of all available views into a single cancer suspiciousness score. While calcifications are marked as in traditional computer-aided detection (CAD) systems, only a small number of soft-tissue lesion marks are shown and are proven to have low false positive rates. However, readers can probe any suspicious image region for decision support to help determine whether further investigation is needed.
The Koios Decision Support (DS) Platform is proprietary and patented cloud-based software built on proven machine learning algorithms. The platform is trained on an ever-growing database of tens of thousands radiologic images linked to pathology results. It is a cloud software that uses AI to assist the radiologist in reading ultrasound images. Koios technology uses a massive database of medical imaging, pathology data and machine learning to provide physicians with a powerful weapon in the fight against cancer. Koios analyzes 17,900 features per image, executes concurrent algorithms trained by hundreds of thousands of images as pathology results.
As technology advances, so do radiologists’ needs. The continuous development and refinement of imaging systems and software can help lead to earlier-stage diagnosis, which in return is what saves lives.
References
1. www.grandviewresearch.com/press-release/global-breast-imaging-equipment. Accessed April 26, 2018.
2. www.businesswire.com/news/home/20170222006174/en/Global-Breast-Imaging-Market-Trends-Analysis-Forecasts. Accessed April 26, 2018.


 May 22, 2026
May 22, 2026 









