
Getty Images
The use of positron emission tomography (PET) imaging in preclinical oncology investigations has shown the ability to determine the efficacy of potential novel treatments. These preclinical cancer studies can improve understanding of the mechanisms of tumor progression, response to cancer treatment and drug toxicity. This article reviews recent developments using PET to evaluate the therapeutic efficacy of drug candidates in preclinical oncology research.
Developing new oncology drug candidates is an expensive endeavor. It is estimated that less than 5 percent of drugs that reach Phase I trials move on to achieve marketing authorization,1 and only 1 in 10,000 pre-clinical compounds ever reach the market.2 Well-designed Phase I trials could be improved by better understanding the dynamic biological processes that may lead the identification of drug candidates with a higher probability of success.
Positron emission tomography is a nuclear molecular imaging technique used in clinical diagnostic and preclinical applications and is often combined with other imaging technologies — such as magnetic resonance imaging (MRI) and computed tomography (CT) scanning — to incorporate anatomical information.
Small animal studies are necessary to explore and validate imaging agents in the preclinical phase, before beginning clinical trials. PET imaging is increasingly being used by researchers in the drug development phase as it provides data that can be translated from animal to human studies. Small animal imaging deepens the understanding of disease development and the effect of potential treatments, and advances in PET technology are powering the translation of this research into the clinical setting.
PET can show the spatial distribution of biomolecules in living tissues, making it particularly useful for evaluating drug candidates in preclinical studies. PET creates three-dimensional (3-D) images of the subject using radioactive tracers — e.g., molecules labelled with a radioactive isotope — usually injected intravenously. The following review outlines recent developments in preclinical oncology research using PET and multi-modal PET imaging.
Therapeutic Efficacy of Drug Candidates
The non-invasive, sensitive and quantitative nature of PET imaging is utilized in preclinical pharmaceutical research to advance knowledge of diseases and drug activity in the body. Multi-modal PET imaging, such as PET/MR and PET/CT, can map the path of drugs throughout the body over time, enabling researchers to monitor efficacy and establish suitability for clinical use.
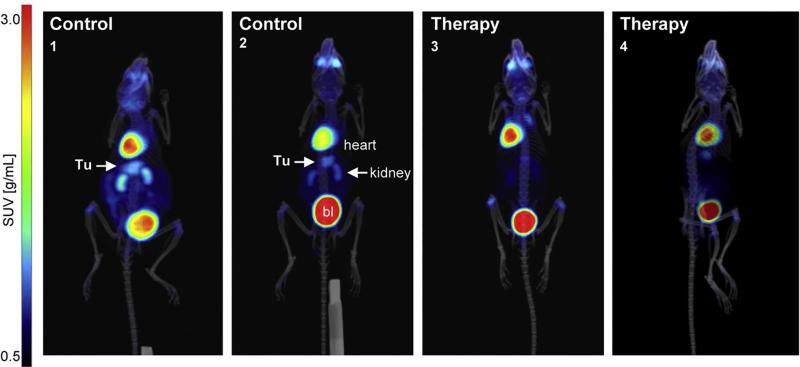
A study using small animal PET/CT imaging confirmed the preclinical efficacy of fasudil — an approved drug for cerebrovascular bleeding — for inhibiting tumor growth in gastric cancer (GC).3 Glucose analogue tracers, such as 18F-fludeoxyglucose (18F-FDG), can monitor glucose uptake in tumors and detect molecular biomarkers to distinguish and quantify tumor burdens.
In this study mice were injected with 18F-FDG and imaged using a small animal PET/CT/SPECT system (Albira PET/SPECT/CT, Bruker BioSpin, Billerica, MA). Quantitative calculation of the 3-D tumor volumes showed a reduction in 18F-FDG uptake and signal intensity by fasudil, compared to non-treated control animals (see Figure 1). As a result, researchers concluded fasudil is a viable novel strategy for GC treatment.
ImmunoPET and Personalized Medicine
Molecular imaging with radiolabeled antibodies, called immunoPET imaging, can provide quantitative information about antibody uptake at a whole-body level.4 ImmunoPET combines PET with an antibody's propensity to home in on the cells it is made to recognize. This imaging technique has shown potential for the assessment of biomarker expression status and/or prediction of clinical response, with a growing number of antibodies being radiolabeled for immuno-PET.5
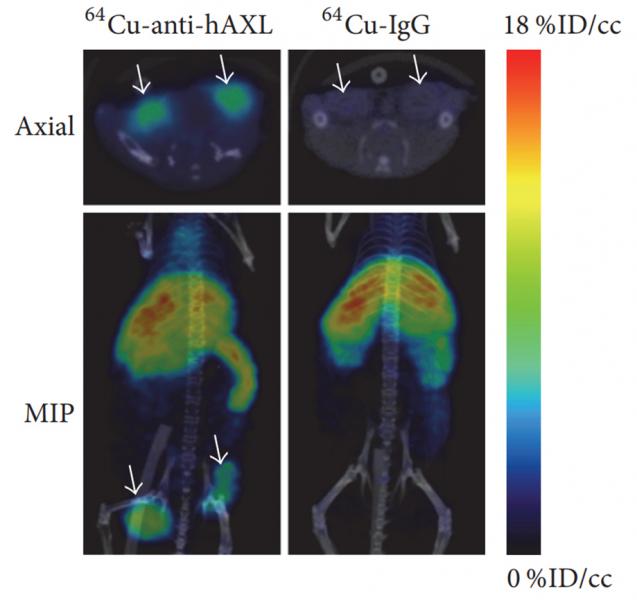
Researchers have used PET/CT for imaging receptor expression in cancer models, for example for treating triple-negative breast cancer (TNBC).6 The heterogeneity of TNBC and lack of actionable targets make treatment challenging, resulting in an unmet clinical need for new molecular targets. The literature suggests that AXL — a member of the receptor tyrosine kinase TAM subfamily — plays a role in TNBC and other cancers, and could be a potential therapeutic target.
Using PET/CT (Albira, Bruker BioSpin, Billerica, MA), expression of AXL and its downregulation by 17-allylamino-17-demethoxygeldanamycin (17-AAG) — a potent inhibitor of heat shock protein 90 (HSP90) — was imaged and quantified using copper-64-labelled anti-human AXL antibody (64Cu-anti-hAXL) as a radioactive probe, shedding light on therapeutic efficacy for AXL-targeted molecular therapies (see Figure 2). Both PET imaging and radionuclide therapy are made possible by the short half-life (~13h) and β+ and β− emissions of 64Cu, making it a desirable PET radionuclide for antibody and nanoparticle labeling.
The in vivo imaging experiment showed that 64Cu-anti-hAXL had greater tumor uptake and accumulation than non-specific 64Cu-IgG, suggesting that 64Cu-anti-hAXL specifically binds to AXL-expressing tumor cells. By using 64Cu-anti-hAXL as an imaging probe, the researchers demonstrated non-invasive assessment of AXL expression using PET/CT in TNBC may be used to predict drug resistance and response to therapies directed at AXL. Such results could aid the development of future theranostic agents, which integrates diagnosis and therapeutics.
Advantages of PET
The non-invasive, sensitive, and quantitative nature of PET imaging enables preclinical researchers to advance knowledge of diseases and drug activity in the body. Advantages offered by PET imaging include high sensitivity for measurement and very low drug concentrations, as well as high biochemical resolution to allow differentiation of physiological alterations that occur nanometers apart.7
As researchers expand the use of multimodal PET imaging in the drug development phase, this data can enable the understanding of cancer-related disease mechanisms, as well as facilitate the development and evaluation of novel therapeutic strategies. Imaging technologies have shown promise in their ability to help establish better preclinical models, which then can improve the possibility of identifying drug candidates with a higher probability of success.

Todd Sasser, Ph.D., is head of applications for Bruker Preclinical Imaging. He works directly with research sites involved in PET applications across a wide variety of disciplines from infection imaging, cancer biology and probe development. Sasser studied at The University of Liverpool and The University of Hawaii and was a visiting scholar at The University of Notre Dame.
References
- Kola I, Landis J. Can the pharmaceutical industry reduce attrition rates? Nat Rev Drug Discov 2004;3(8):711-15.
- Tonkens R. An overview of the drug development process. Physician Exec 2005;31(3):48-52.
- Hinsenkamp I, Schulz S, Roscher M, Suhr A, Meyer B, Munteanu B, Fuchser J, Schoenberg SO, Ebert MPA, Wängler B, Hopf C and Burgermeister E (2016) Inhibition of Rho-Associated Kinase 1/2 Attenuates Tumor Growth in Murine Gastric Cancer, Neoplasia, 18;500-511.
- van Dongen GAMS, Visser GWM, Lub-de Hooge MN, de Vries EG, Perk LR. Immuno-PET: a navigator in monoclonal antibody development and applications. Oncologist. 2007;12:1379–89.
- Alauddin MM, Khawli LA. Advances in Immuno-PET in the Detection of Cancer and Assessment of Response to Therapy. Curr Med Chem. 2020 Jan 27.
- Wang W, Zhao J, Wen X, Lin CC, Li J, Huang Q, Yu Y, Lin S and Li C (2017) MicroPET/CT Imaging of AXL Downregulation by HSP90 Inhibition in Triple-Negative Breast Cancer, Contrast Media & Molecular Imaging, 2017, Article ID 1686525.
- Fischman AJ, Alpert NM and Rubin RH (2002) Pharmacokinetic Imaging: A Noninvasive Method for Determining Drug Distribution and Action, Clin Pharmacokinet, 41(8): 581-602.


 May 08, 2026
May 08, 2026 









