Image courtesy of Siemens Healthineers
There have been a few big, recent advancements in cardiac computed tomography angiography (CCTA) imaging technology. The biggest of these was the introduction of two CT systems aimed specifically at the coronary imaging market — the GE Healthcare Cardiographe dedicated cardiac CT scanner and the Canon (formerly Toshiba) Aquilion Precision.
The Cardiographe was developed by Israeli start-up company Arineta with input from several luminary physicians. It incorporates numerous technologies that are specifically aimed at improving coronary CT angiography, without concerns about using the machine for general radiology imaging. The company partnered with GE Healthcare to commercialize the scanner, which gained U.S. Food and Drug Administration (FDA) clearance in 2017. GE incorporated many of its latest CT advances for dose reduction, iterative reconstruction, metal artifact reduction and other features from its Revolution CT system. The Cardiographe was built to be very compact so it can fit into a physician’s office, or utilize a smaller room than a standard 64-slice scanner. It offers 140 mm of anatomical coverage to scan the entire heart in one rotation without stitching. Its gantry rotation speed is 0.24 seconds, enabling very fast scans that freeze cardiac motion. Examples of CTA scans on the system presented in sessions were performed at 2 mSv of dose or below.
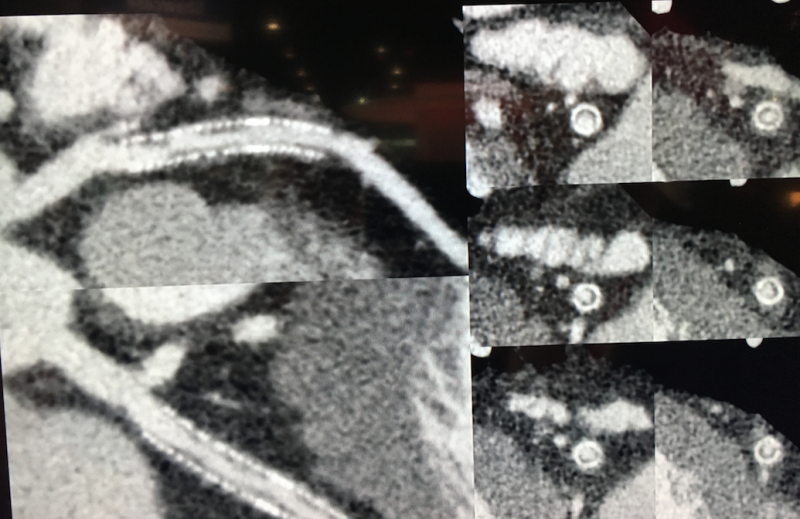
The Aquilion Precision was unveiled for the first time at the 2017 Radiological Society of North America (RSNA) meeting and was cleared by the FDA in April 2018. It is unique in that it is the first CT system to offer a 0.25 mm resolution, rather that the standard 0.5 mm resolution on most of today’s scanners. It is capable of resolving coronary anatomy as small as 150 microns, providing CT image quality with resolution typically seen only in cath lab angiography. This enables better visualization of tiny structures, such as the composition of coronary plaques, stent struts and smaller coronary vessels that are generally difficult to see on CTA scans. The resolution may aid in better plaque assessments, including clearer identification of vulnerable plaques, visualizing broken stent struts, and identification of a specific vendor’s stents based on strut design in vessels.
The Precision uses a newly designed detector to provide more than twice the resolution of 0.5 mm systems. The scanner offers dose efficiency with detector channels that are only 0.25 mm thick. It also has improvements in scintillator quantum efficiency, detector circuitry and other components. The system features the industry’s smallest focal spot tube, at 0.4 x 0.5 mm, and the industry’s first routine 1,024 x 1,024 reconstruction matrix.
Key Specifications for CT Cardiac Imaging
Here is a list of some key technologies that should be considered when evaluating new CT scanners for cardiac imaging.
1. Speed — The faster the temporal resolution a scanner has, the faster its gantry can rotate around a patient. Some of the newest scanners move fast enough to capture images that freeze cardiac motion and prevent motion blur or the need to stitch images from several heartbeats to reconstruct a complete cardiac image.
2. Motion correction algorithms — This software can help compensate for cardiac motion to provide clearer images. This can help reduce the need for retakes and help reduce patient dose.
3. Cardiac metal artifact reduction software — Many cardiac patients have implanted hardware from previous procedures, including stents, surgical staples, sternum wires, septal or left atrial appendage (LAA) occluders, and pacemaker or implantable cardioverter defibrillator (ICD) leads. Any of these metal implants can cause metal artifact blooming or streaking artifacts that compromise the diagnostic quality of the images. The latest generation software offered by most vendors can largely compensate the photon starvation behind the metal or can reduce and cut through glare to see the underlying anatomy. Some newer software can increase image quality enough to visualize plaques or clots inside stents.
4. Calcium scoring software — Data continues to mount that calcium scoring is a good predictor of a patient’s risk for future coronary events. It is also being advocated for screenings of patients who are on the fence about going on statin therapy.
5. Plaque assessment software — This software is offered by all of the top CT system vendors and most of the third-party advanced visualization vendors. As more research accumulates and image resolution on CT scanners improves, quantifying the types of plaques seen in the coronary arteries on CT will likely play a bigger role. These CT assessments can help determine if an intervention is needed and help guide those interventions.
6. CT perfusion — This software tracks the iodine contrast concentration levels in the myocardium. The software reconstructs color coded maps showing areas of low concentration (areas of poor blood flow caused by ischemia), similar to nuclear myocardial perfusion exams that show areas of poor metabolic activity due to ischemia. The data from these exams is available almost immediately, whereas nuclear scans may require the patient to be admitted overnight.
7. TAVR / structural heart planning software — CT systems with workflows optimized for transcatheter aortic valve replacement (TAVR) or other structural heart exams will be helpful as these procedures continue to see rapidly increasing volumes. Software for TAVR, mitral valve assessments, LAA and septal defect occlusion planning should be considered in health systems offering these procedures.
8. FFR-CT assessment capability — As fractional flow reserve computed tomography (FFR-CT) gains reimbursement it is expected to see wider adoption. It can help definitively assess chest pain patients and either rule out coronary artery ischemia, or pinpoint the culprit lesions and the severity of blood flow obstruction, which can help better guide interventions. Invasive catheter-based FFR uses blood flow pressure readings before and after a coronary lesion to determine if a stenosis is hemodynamically significant and requires stenting. FFR-CT is noninvasive and uses super-computing, computational fluid dynamics to assess a virtual FFR, which may help eliminate the need for diagnostic catheterization.
For more insights on what to look for in cardiac CT scanners, watch the VIDEO “What to Consider When Comparing 64-slice to Higher Slice CT Systems,” an interview with Claudio Smuclovisky, M.D..
Other New CT Scanners on the Market
GE unveiled a premium CT with similar specs, the Revolution Frontier, which has cleared the FDA and is expected to begin shipping in 2Q 2018. It is built around the company’s Gemstone Clarity detector and Performix HD Plus X-ray tube. The super-premium CT can visualize structures to a resolution of 0.23 mm with 25 percent less electronic noise, according to GE. It also features spectral imaging.
Siemens extended the concept of patient-centric mobile workflow with two new members of its “go” family – the Somatom go.All (64 slices per rotation) and go.Top (128 slices per rotation). The systems use a tablet device and remote controls, to eliminate the need for a traditional control room and enable more time with the patient scanner side.
The 64-slice go.All can scan up to 100 mm in one second. The 128-slice go.Top can cover up to 175 mm per second with a range of 200 cm, enough to scan the length of a patient more than 6 and a half feet tall. The X-ray tube can be adjusted in 10-kilovolt increments.
The go.Top also offers TwinBeam Dual Energy imaging. This splits the X-ray beam into two energy spectra before it reaches the patient, enabling providers to simultaneously examine the same body region at two different energy levels.
FFR-CT Can Identify Lesions Requiring Stenting
One of the new add-on advanced visualization technologies for CCTA that vendors are now offering is fractional flow reserve computed tomography (FFR-CT). It offers a completely a noninvasive FFR assessment of not just a coronary segment, but the entire coronary tree. Invasive catheter-based FFR uses blood flow pressure readings before and after a coronary lesion to determine if a stenosis is hemodynamically significant and requires stenting. Traditionally CTA has been great at showing anatomical detail, including the percentage obstruction a lesion presents. But in the cath lab, it is often the case where a significant blockage seen on CT does not greatly impact blood flow. To avoid the need for a diagnostic catheterization for confirmation, FFR-CT uses super-computing computational fluid dynamics to assess a virtual FFR, which studies have shown has an 80 percent correlation with invasive FFR. This may be the key in making CT a true gatekeeper to the cath lab and for EDs to more quickly discharge chest pain patients where coronary blockages can be ruled out.
Several vendors have recently created partnerships with HeartFlow, which offers FFR-CT technology that is cleared by the FDA.
As CCTA sees wider adoption in emergency departments for quicker evaluation of chest pain patients, FFR-CT will likely see an increasing role to verify patients requiring percutaneous coronary intervention (PCI) and help reduce or eliminate the need for invasive diagnostic angiograms.
Comparison Chart of Cardiac CT Systems
This article served as an introduction for a cardiovascular CT system comparison chart in the May-June 2017 print issue of DAIC. To access the chart, go to www.dicardiology.com/content/cardiac-ct-systems. This will require a login, but it is free and only takes a moment to complete the form. This also will grant access to more than 40 other comparison charts.
Vendors in this chart include Canon, GE Healthcare, NeuSoft, Philips Healthcare and Siemens Healthineers.
Related Cardiac CT Content:
Advances in Cardiac Imaging Technologies at RSNA 2017
Advances in Cardiac CT Technology
New Developments in Cardiovascular Computed Tomography at SCCT 2017
VIDEO: Editor's Choice of Most Innovative New Cardiac CT Technology at SCCT 2017
VIDEO: Role of Cardiac CT in Value-based Medicine
VIDEO: Latest Trials on Cardiac CT Compared to Functional Testing
VIDEO: Clinical Evidence Shows CT Should be the Primary Cardiac Imaging Modality
VIDEO: Advances in Cardiac CT Imaging
VIDEO: The Future of Cardiac CT in the Next Decade, an interview with Leslee Shaw, Ph.D., Emory.
VIDEO: What to Consider When Comparing 64-slice to Higher Slice CT Systems, an interview with Claudio Smuclovisky, M.D.


 April 29, 2026
April 29, 2026 









