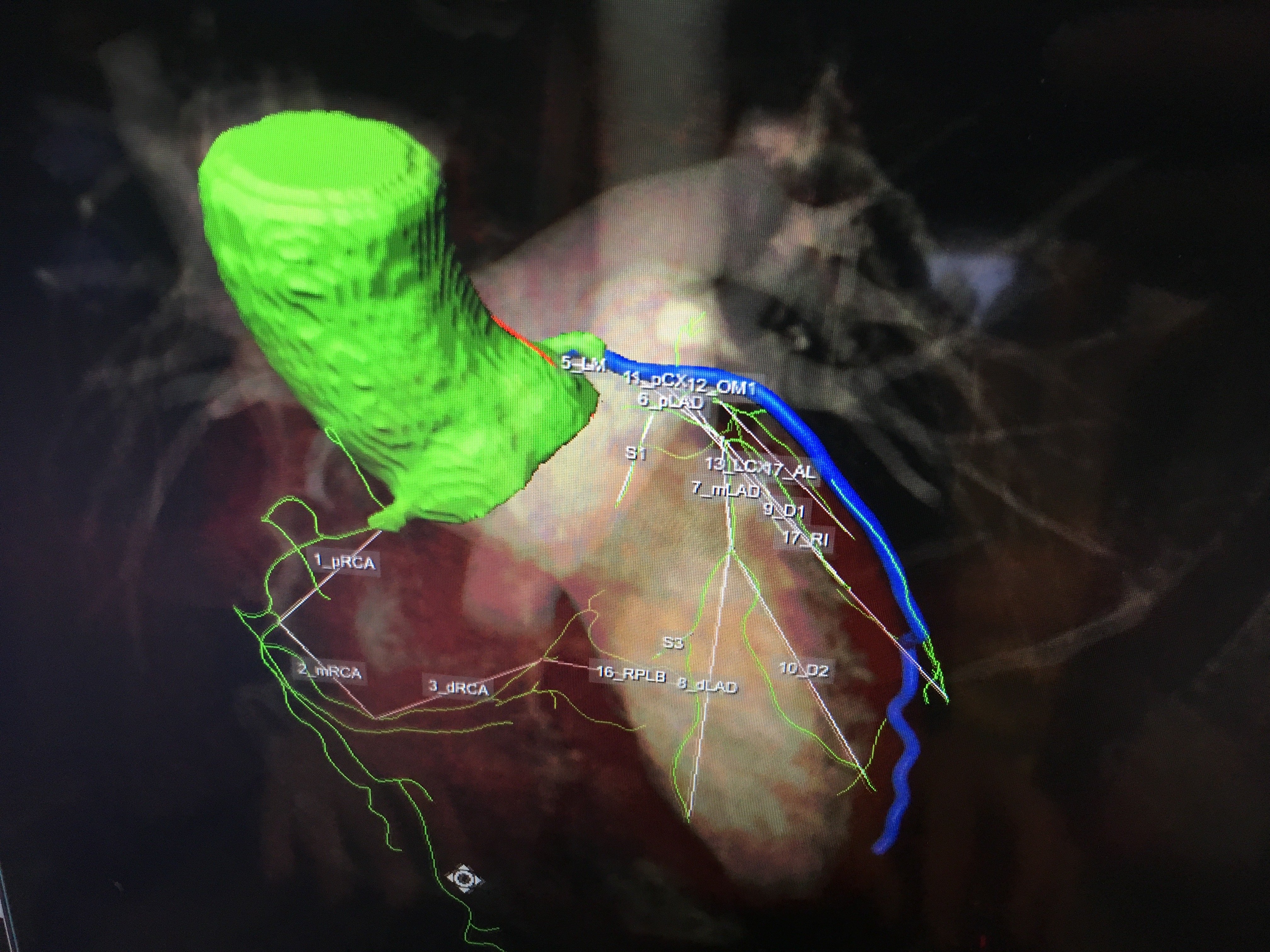
An example of the newest generation of smart cardiac CT software that automatically identifies the anatomy, autotraces the centerlines on the entire coronary tree and labels each vessel segment. This greatly speeds CT workflows, saving time for techs, radiologists and cardiologists.
Here is a checklist of dose-sparing practices for cardiac computed tomography (CT) imaging used in the cath lab. This list was included in a 2018 consensus document to guide the optimal use of ionizing radiation in cardiovascular imaging.1
The consensus document was issued in May 2018 jointly by the American College of Cardiology (ACC), Heart Rhythm Society (HRS), North American Society for Cardiovascular Imaging (NASCI), Society for Cardiovascular Angiography and Interventions (SCAI), Society of Nuclear Medicine and Molecular Imaging (SNMMI) and the Society of Cardiovascular Computed Tomography (SCCT).
It includes input from experts from a total of nine cardiology societies, and includes best practices for safety and effectiveness when using computed tomography (CT), nuclear imaging and angiographic/fluoroscopic imaging.
One section includes the following checklist to help lower dose in cardiovascular CT angiography (CCTA):
Case Selection:
• Consider patient age, co-morbidities, natural life expectancy; and
• Consider appropriateness and utility of nonradiation-based imaging techniques.
Equipment Calibration:
• Use acquisition detector doses as low as compatible
with diagnostic image quality.
Procedure Planning:
• Select lowest-dose acquisition protocol compatible with study goals;
• Use ECG-gated variable tube output if retrospective gating is used;
• Use the lowest X-ray tube voltage compatible with adequate diagnostic-quality image acquisition;
• Use the lowest X-ray tube current compatible with diagnostic-quality image acquisition; and
• Use the largest scan pitch compatible with adequate diagnostic-quality image acquisition.
Study Conduct:
• Minimize patient heart rate; and
• Confine scanned body area to the area relevant to the study’s diagnostic purpose.
Variables That Affect Patient Dose for X-ray CT
The radiation dose to a patient is determined by a combination of the patient’s physical characteristics and scanner protocol selection. Patient exposure will necessarily increase with patient size and body mass index. Depending on the specific acquisition parameters, the increased exposure need not increase dose to radiosensitive tissues. Patient size is not a variable that determines exam appropriateness as long as the patient’s size does not preclude obtaining diagnostic-quality images.
X-ray CT systems can either use a constant X-ray tube output or, in some acquisition protocols, use ECG-gated variable output. The operator selects the acquisition protocol based on patient characteristics and the study purpose, with the intent to deliver a sufficient exposure to permit an acceptable degree of noise in the reconstructed images.
The X-ray CT system operator is responsible to select the scanning protocol that optimizes the examination’s diagnostic yield while minimizing dose. The following are essential considerations in this process:
1. Scan length. Scan length, defined as the distance imaged along the cranio-caudal axis, should be kept to a minimum to encompass only the anatomy of interest and not expose structures that are not relevant to the examination’s purpose. Care needs to be taken to ensure that the diaphragm position seen on the topogram is the same as during the scanning. This requires similar breath-hold instructions.
2. X-ray beam intensity. X-ray beam intensity is determined by both the X-ray tube potential (in units of kV) and the X-ray tube current (in units of mA). Modern
CT scanners modulate the tube current dynamically throughout the CT acquisition to minimize radiation exposure.
Tube potential: Studies of radiation dose reduction have demonstrated that the most important single factor in controlling radiation dose is adjustment of X-ray tube voltage. Increasing tube voltage increases the X-ray beam’s mean photon energy level, and increases radiation dose roughly proportionally to the square of the voltage. Thus, at a constant tube current, a decrease of tube voltage from 120 to 100 kV reduces the radiation dose by almost 40 percent. In most scanners, the X-ray tube voltage may be adjusted between
70 to 140 kilovolts (kV). The voltage is selected by the operator based on subject weight or body mass index. A commonly used adjustment scale that provides diagnostic quality in most scanners is: 120 kV for patients with body mass index ≥30 kg/m2, 100 kV for body mass index 21 to 29 kg/m2, and 80 kV for body mass index <21 kg/m2. Image noise decreases as potential increases, so that in extreme cases (body mass index ≥40 kg/m2) the maximum tube potential of 150 kV may be necessary
to produce diagnostic-quality images.
Tube current: The X-ray tube current (in mA) is defined as the number of electrons accelerated across the tube per unit of time and is proportional to the number of X-ray photons produced per unit time. The radiation dose is linearly proportional to the tube current. Image noise is inversely proportional to the square root of the tube current. Thus, decreasing tube current at a given tube potential decreases the radiation dose at the expense of increased image noise. The tube current may be modified based on patient size assessed by visual inspection, measurement of body weight or body mass index, thoracic circumference or diameter, or noise measurement from a cross-sectional prescan or topogram. Most modern scanners offer tube current modulation based on the thickness of the body estimated from the topogram. Modulation may be applied longitudinally as well as circumferentially. This approach can reduce radiation exposure of thoracic CT examinations by 20 percent without increasing image noise.
3. Rotation time. The time required for the gantry to perform one rotation is a selectable parameter. Exposure increases linearly with rotation time.
4. X-ray beam filtration. Filters placed beneath the X-ray tube are used to selectively attenuate low-energy X-rays that do not significantly contribute to the image but do contribute to radiation dose. The net effect is to increase the mean energy of the X-rays while not altering the maximum energy.
5. Scan acquisition mode. This is a major determinant of radiation dose. Different acquisition modes can deliver substantially different doses while producing similar images. There are three principal CT scan modes: axial or “conventional” scanning, helical scanning, and fixed-table or single-station scanning.
6. Cardiac motion compensation. Compensation for cardiac motion is rarely applied outside of direct cardiac and aortic root imaging. Thus, the majority of cardiovascular medical imaging does not employ ECG gating or triggering. In contrast, when imaging the heart or aortic root, cardiac motion compensation is critical to avoiding motion-related artifacts that substantially degrade image quality. Depending upon the scan mode, one of two cardiac compensation methods is used:
Prospective ECG triggering: Prior to the scan, the operator “prospectively” selects an imaging window within the cardiac cycle, which may be defined as a percentage from one R-wave to the next, or an absolute time delay after each R-wave. Scans are then triggered to coincide with the selected scan window. Prospective triggering may be applied to each of the three scan modes. In the case of axial scanning, ECG triggering is used to trigger the acquisition at each table position.
Retrospective gating: Applicable to both helical and fixed table scan modes. With helical scanning, acquisition is performed using a low pitch of approximately 0.2. The slow acquisition images the entire cardiac anatomy across the entirety of a cardiac cycle, providing a 4-D dataset that allows each spatial location within the heart to be reconstructed at any time-point across the cardiac period. Data is continuously acquired along with the ECG signal while covering the anatomy of interest. The data is subsequently rebinned at each slice location for image reconstruction, according to the time of the cardiac cycle from the ECG signal.
ECG-triggered tube current modulation: As discussed in the preceding section, “tube current,” ECG-triggered tube current modulation is used to reduce radiation dose during systole when there is the greatest cardiac motion and can reduce the radiation exposure significantly. In this circumstance, tube current is at nominal value only during the portion of the cardiac cycle likely to be used for reconstruction (typically end diastole). During the remainder of the cardiac cycle, the tube current is reduced to minimize radiation output. Recent refinements of this technique have allowed reduction of the length of time (“window”) during which tube current is nominal and reduction of tube current during the undesired portions of the cardiac cycle by 20 percent and to as little as 3 to 5 percent of the nominal value. A potential disadvantage of this technique is that images reconstructed from projection data acquired with low tube current may be too noisy to be diagnostic for coronary anatomy. Retrospectively ECG-triggered tube current modulation works best in patients with stable sinus rhythm and low heart rates (specific thresholds depend on scanner characteristics).
7. Image reconstruction. Filtered back-projection has historically been used to reconstruct CT images from projection data. The advent of greater computing power has made an alternative statistical method — iterative reconstruction — practical for CT. This method predicts projection data based on an initial assumption about the attenuation in each voxel, and compares that data to measured projection data. The voxel attenuation values are modified iteratively until an acceptable level of error between the predicted and measured data is obtained. The resulting reconstructed images have lower noise values compared with those obtained with filtered back projection. This permits reducing tube voltage and/or current to obtain images with comparable noise and lower radiation dose. One important characteristic of iterative reconstruction is that excessively low-dose images do not appear grainy, as is the case with filtered back projection. Instead, structures become blurred and can develop a blotchy appearance, undermining their diagnostic effectiveness.
8. Image post-processing filters. These may also be applied to acquired images to reduce image noise while preserving image contrast and edges. The feasibility of using these filters for radiation dose reduction has been recently demonstrated.
Reference:
1. John W. Hirshfeld Jr., Victor A. Ferrari, Frank M. Bengel, et al. 2018 ACC/HRS/NASCI/SCAI/SCCT Expert Consensus Document on Optimal Use of Ionizing Radiation in Cardiovascular Imaging: Best Practices for Safety and Effectiveness: A Report of the American College of Cardiology Task Force on Expert Consensus Decision Pathways. Journal of the American College of Cardiology. May 2018. DOI: 10.1016/jacc.2018.02.016.


 May 05, 2026
May 05, 2026 









