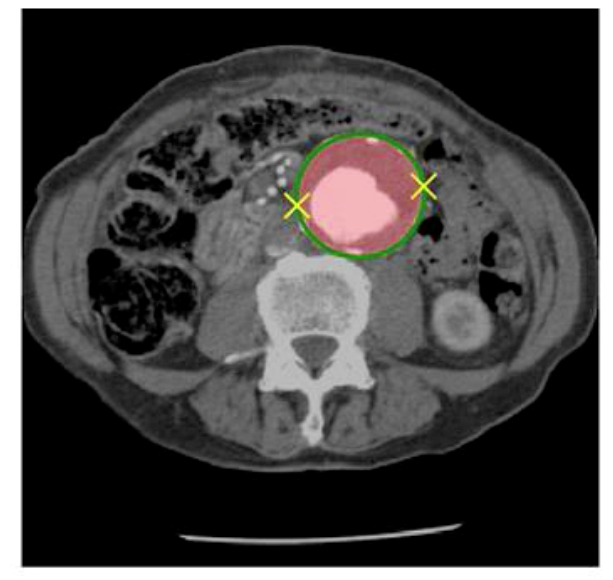
The DeepAAA algorithm, developed at the MGH & BWH Center for Clinical Data Science, accurately detected and measured an abdominal aortic aneurysm (AAA) in a CT image even though appearance of the AAA was complicated by a blood clot. (The algorithm drew a green circle around the aneurysm.) Image courtesy of Varun Buch, MGH & BWH Center for Clinical Data Science
Editor’s note: This article is the second in a content series by Greg Freiherr covering the Society for Imaging Informatics in Medicine (SIIM) conference in June.
Aneurysms of the abdominal aorta may occur in elderly men. They can burst suddenly, killing more than 90 percent. Yet abdominal aortic aneurysms (AAAs) typically are asymptomatic. Often they are found during imaging exams performed for other reasons.
This tendency toward incidental discovery in the context of their prevalence and potential lethality led to the development of DeepAAA, an algorithm that uses artificial intelligence (AI) to search pelvic and abdominal CTs for signs of an abdominal aortic aneurysm.
“The idea (behind DeepAAA) is that if you’re using an algorithm that can work automatically in the background, you can screen for these aneurysms automatically,” said Mark H. Michalski, M.D., executive director of the Center for Clinical Data Science, which is operated jointly by the Massachusetts General Hospital (MGH) and Brigham and Women’s Hospital (BWH) in Boston.
Spotting patients at risk of complications from a burst abdominal aneurysm is one — but not the only — reason Michalski and his team at the MGH & BWH Center for Clinical Data Science developed DeepAAA. Another is to understand why AAAs only threaten the lives of some patients with this type of aneurysm.
“Sometimes it never progresses. Sometimes it progresses fast,” he said. “Understanding early who will progress would be really useful.”
SIIM presentation
DeepAAA has successfully identified potentially troublesome abdominal aortas, even when blood clots made discovery of the widening blood vessel difficult, Michalski said. The MGH/BWH team plans to formally report their findings during a scientific session Friday, June 28 at the annual meeting of the Society for Imaging Informatics in Medicine (SIIM).
DeepAAA is already in use at MGH and BWH to automatically search for AAAs. The resulting information may signal needed patient follow-up. It may also identify risk factors. Research on these factors may lead to the discovery of biomarkers “that can tell you what kind of patients are likely to rupture or which patients are most likely to progress,” Michalski said.
Currently known risk factors include age (elderly), gender (male); cigarette smoking, and hypertension. “But (an aneurysm) can happen to anyone,” he said.
Productizing AI Algorithms
The algorithm might have commercial prospects. The Center for Clinical Data Science is exploring strategic alliances with companies that might productize DeepAAA, according to Michalski, who explained that the Center seeks to demonstrate the potential of the AI algorithms, then hopes to hand them off to companies for productization.
“Partnerships with them are really important,” he said. “We don’t intend to build a sales force and we can’t be sure of the quality of these models across multiple sites. That is what the vendors do very effectively.”
DeepAAA is just one of several AI-infused algorithms being developed by the center. Others automatically check head CTs for stroke or hemorrhage. None is perfect, Michalski said. “But because we are at a big academic medical center, people understand that and they can help us improve these models on-the-fly.”
In the coming weeks, a software team at the center will begin making the adjustments that will fit these algorithms into clinical workflows, the metaphorical equivalent of fitting handles to shovels.
These “handles” might allow the AI-algorithms to be used in PACS or in CT scanners dedicated to hospital emergency departments, according to Michalski. The algorithms must also be integrated into the reporting or ordering systems “to help integrate (findings) into your report and to prompt you to do follow-up exams,” he said.
But many challenges remain. One involves training. When training DeepAAA, for example, developers must consider that abdominal aortas look different in different people. Some have a lot of curves; others have calcifications. And structures around and in the aorta (e.g., blood clots) can complicate automatic measurement of the blood vessel perimeter, which is essential if the early signs of AAAs are to be identified.
“The process by which we created this model (DeepAAA) — and validated it across several hundred additional and different cases — showed us that indeed the model works and can perform well,” he said.
Proven Feasibility
Michalski noted to Imaging Technology News (ITN) that this only proves “feasibility.” Productization may require the inclusion of a “function” at the clinical interface that allows the model to be tuned to patient populations other than the ones at the large academic center where DeepAAA was developed. This function may allow the addition of data representative of local patient populations, Michalski said.
Such additional training, which will involve deep learning, may take only a few months. But the additional images would have to be clearly labeled as indicating the presence of AAA.
In the meantime, the center is expanding the mission of DeepAAA to the thorax to look for signs of aortic aneurysms in chest CTs. Although these aneurysms occur less frequently than AAAs, they are also silent and, if they burst, can lead to medical complications or death.
Greg Freiherr is a contributing editor to Imaging Technology News (ITN). Over the past three decades, he has served as business and technology editor for publications in medical imaging, as well as consulted for vendors, professional organizations, academia, and financial institutions.
Related content:
Making AI Safe, Effective and Humane for Imaging
How Artificial Intelligence Can Predict and Detect Stroke
Novel Artificial Intelligence Method Predicts Future Risk of Breast Cancer


 May 22, 2026
May 22, 2026 









