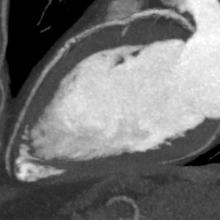
August 14, 2014 — Toshiba Medical Systems Europe will introduce the SURE Subtraction Coronary software at the 2014 European Society of Cardiology (ESC) Congress in Barcelona, to be held Aug. 30 to Sept. 3.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now