
As the nascent market for artificial intelligence (AI) in medical imaging develops, healthcare providers are faced with the question of how best to select, purchase and implement the rapidly expanding range of regulatory approved AI-enabled imaging applications and AI algorithms.
More than 50 vendors have received regulatory clearance for medical imaging AI products and with more than 200 algorithm developers active in the market, product availability will increase many-fold in the coming years. Purchasing algorithms direct from multiple vendors adds an administrative overhead, not to mention the technical challenges of integrating these algorithms in existing systems and clinical workflows. AI platforms and marketplaces, akin to mobile app stores, offer a unified approach to deploying AI in medical imaging. A summary of the advantages and disadvantages for healthcare providers of AI marketplaces is provided in Figure 1.
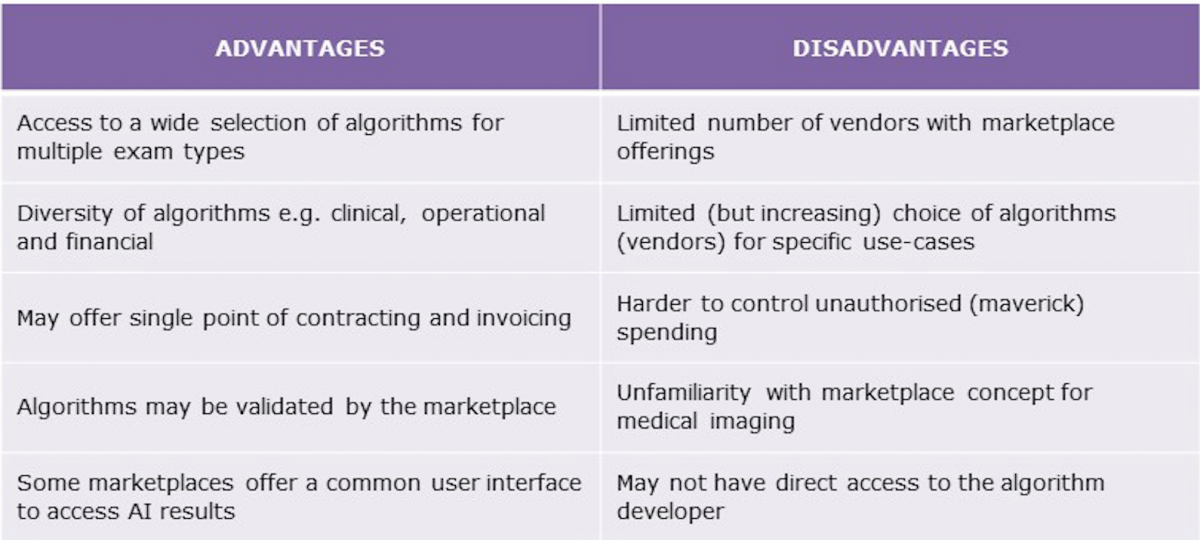
The first marketplace/platform offerings entered the market in the last couple of years, and several new ones were announced at the 2019 annual meeting of the Radiological Society of North America (RSNA 2019). The performance and functionality of these solutions varies between vendors, with some offering what is essentially an online application store, with little or no support for integration, and others offering end-to-end solutions that address many of the engineering (deployment, workflow integration, etc.) and commercial (contracting, billing, etc.) challenges associated with the distribution and implementation of AI.
Selecting a Vendor
Some of the criteria that healthcare providers should consider when selecting a marketplace/platform vendor includes partnerships, applications, regulatory clearance, workflow, functionality, deployment, contracting and support. This selection guide is based on verified contributions from each of the vendors. Signify Research market data analysts anticipate this will be a useful resource for healthcare providers when determining whether to implement a marketplace offering, and then, which vendor to select. You can view the complete flowchart at https://bit.ly/2TR7RC8.
AI marketplaces with the most partners and greatest number of applications and algorithms may, at first sight, appear the obvious to partner with. However, healthcare providers are advised to consider how these will be implemented in clinical practice, and the level of contracting and technical support provided by the marketplace. The selection guide (see above link) also covers a range of other criteria and functionalities that should be considered, and how today’s marketplaces perform against these criteria. For example, when considering workflow integration, providers are advised to consider:
• How images are routed to the AI;
•How algorithms/applications are prioritized;
•How the radiologist engages with the AI results; and
•How the results are populated in the radiologist report.
Other considerations include deployment options and tools for clinicians to develop their own AI solutions.
Sanjay M Parekh, Ph.D., is a senior market analyst at Signify Research Ltd. His current research focuses on the artificial intelligence and machine learning in medical imaging market. Signify Research is an independent supplier of market intelligence and consultancy to the global healthcare technology industry. Its major coverage areas include healthcare IT, medical imaging, and digital health. Clients include technology vendors, healthcare providers and payers, management consultants and investors. The company is headquartered at Cranfield, U.K.



 May 12, 2026
May 12, 2026 









