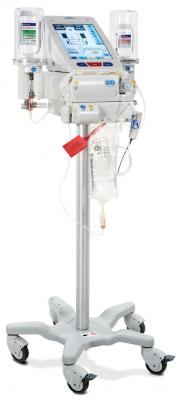
December 5, 2016 — Bracco Diagnostics Inc. announced that CT Exprès — the first and only multi-dose, multi-patient, syringe-less injector for computed tomography (CT) according to Bracco — is now available in the United States. The CT Exprès 3-D Contrast Media Delivery System is designed to streamline workflow and optimize patient care within the CT department.
According to Bracco Diagnostics President and CEO Vittorio Puppo, the volume of CT exams continues to grow, and imaging centers and radiology departments need to control costs, maintain productivity and preserve workflow efficiency. CT Exprès is designed to significantly increase the number of scans clinicians can perform in a day, while delivering a more user-friendly experience and delivering the same high-quality imaging results as dual-syringe systems.
CT Exprès was cleared by the U.S. Food and Drug Administration (FDA) for controlled automatic administration on the venous side, of contrast media and saline. The system is used in combination with Isovue (lopamidol injection) Imaging Bulk Package (IBP) and saline (0.9 percent sodium chloride injection USP). Use of saline with this contrast medium injector allows multi-dosing from a saline container.
In a prospective study of 275 consecutive outpatients, the utility of CT Exprès was compared with a dual-syringe system. The comparison was carried out in a single center utilizing two scanner rooms, which shared a common console room. Patients and technologists were randomized between the rooms, and two radiologists evaluated image quality. Results showed that CT Exprès improved throughput and provides added benefits over dual syringe-based systems including:
- 65 percent less preparation time;
- 75 percent less releasing time;
- Saved over 2 minutes/exam and increased throughput by 2.6 patients/day;
- Reduced contrast media waste;
- Increased technologists' satisfaction; and
- Equivalent image quality.
For more information: www.imaging.bracco.com/us-en


 May 19, 2026
May 19, 2026 









