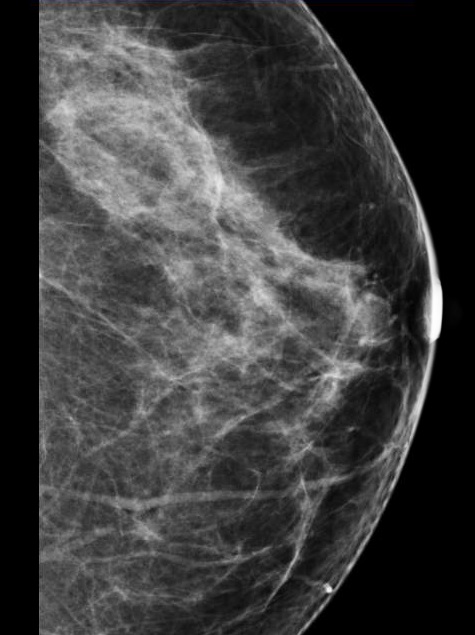
New York State is taking the most aggressive action in the nation to improve access to breast cancer screening with a new law requiring health insurance to cover all screening and diagnostic imaging exams for the detection of breast cancer. The law includes supplemental imaging for all women. The law, signed by Gov. Andrew Cuomo June 27, removes insurance barriers for all women, including cost-sharing in the form of annual deductibles or coinsurance. It is the first law in the country requiring insurance companies to cover all of the costs of supplemental imaging to encourage women with dense breasts to seek additional screenings.
"Eliminating these insurance barriers will prevent women from paying out-of-pocket for breast cancer screening, including imaging for the detection of breast cancer, diagnostic mammograms, breast ultrasounds, or magnetic resonance imaging (MRI),” stated a press release issued by the governor’s office.
The new law (insurance bill S8093) goes into effect Jan. 1, 2017.
In 2013, New York was the first state to adopt a dense breast notification law to inform patients in clear, unambiguous language if their mammogram showed high fibroglandular densities, said New York advocate and Executive Director of DenseBreast-info.org, JoAnn Pushkin. She said after New York's law went into effect, two things became evident. The first was that further education was needed for both patients and their referring physicians about the implications of dense tissue. The second was that uncertainty over insurance coverage and high co-pay/deductibles could be deterrents to follow-up screening using breast ultrasound or MRI, which can see through dense breast issue.
One caveat on the new insurance bill, which it is not specific to New York, is that employer plans set up as "self-funded" are generally Exempt from state insurance laws. This means coverage might still be extended, but patient would have to pay co-pay and deductible.
For more information: DenseBreast-info.org
Related Breast Density Content:
New Technology and Clinical Data in Breast Imaging
Additional Resources on ITN for Breast Imaging and Dense Breast technology Information
Mammographic Breast Density — What It Means
New Technology and Clinical Data in Breast Imaging (articles from 2017)
VIDEO: Advances and Trends in Breast Imaging
Dense Breast Tissue: Supplemental Imaging
Breast Density: Are You Informed?


 May 07, 2026
May 07, 2026 









