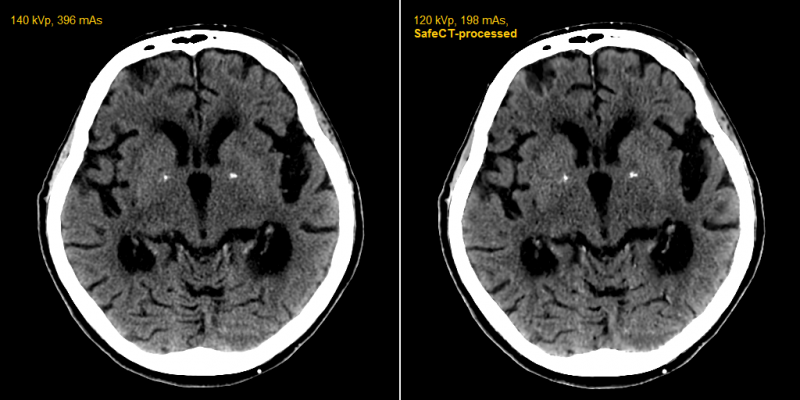
April 16, 2012 — Medic Vision Imaging Solutions Ltd. announced that within six months of clinical use in the United States, its SafeCT image enhancement system has delivered diagnostic image quality to more than 20,000 CT (computed tomography) studies acquired with low-dose protocols.
Using proprietary patented iterative volumetric image reconstruction algorithms, SafeCT delivers diagnostic image quality to studies acquired over a wide range of exposure parameters on any CT scanner. Its use eliminates the need to replace existing CT systems with newer and more expensive scanners to achieve similar results.
SafeCT is currently being used in major hospitals such as University of Pittsburgh Medical Center, Massachusetts General Hospital and Cedars-Sinai, as well as in independent medical imaging practices. SafeCT is installed on networks serving CT scanners ranging from four-slice to 64-slice models from GE, Siemens, Toshiba and Philips.
A single SafeCT can simultaneously serve multiple scanners in different locations. Its use is virtually transparent to department workflow. The noisy images are automatically sent from the CT console to SafeCT, which processes them and transfers the enhanced images in real time to the picture archive and communications system (PACS) and/or workstation without any intervention by the technologist.
“Medic Vision has taken a new approach to achieving diagnostic images from low dose scans so that no equipment replacement, enormous capital budgets, workflow adjustments or applications training are required,” said Eyal Aharon, Medic Vision CEO. “Our sophisticated volumetric algorithms deliver the high diagnostic quality images that radiologists demand.”
For more information: www.medicvision.com


 June 01, 2026
June 01, 2026 









