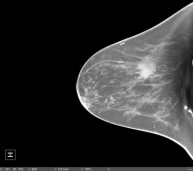
An example of KBCT
March 2, 2012 — Koning Corp. a developer of cone beam computed tomography (CBCT) announced that they obtained CE mark approval for their Koning Breast CT (KBCT) system. This approval signals compliance with relevant European Union (EU) legislation and medical device regulations allowing it to be marketed and sold throughout the EU and other countries recognizing the CE Mark.
"This CE mark approval represents a major step for breast-imaging, and women's health care, in general," said Ruola Ning, Ph.D., Koning's president and founder. "KBCT is the latest advancement in true 3-D breast-imaging technology and focuses on a growing worldwide women's health issue – the early detection and diagnosis of breast cancer. Breast cancer affects hundreds of thousands of women and is one of the most common forms of cancer in North America, Europe and China. We are very proud to be able to bring this revolutionary technology to benefit women throughout the world."
KBCT is the first commercially available, fully-integrated, dedicated breast CT scanner designed specifically to image the entire breast with high spatial and contrast resolution at a radiation dose similar to or less than diagnostic mammography. The system acquires a set of images in ten seconds producing 'true' 3-D images and multi-planar cross-sectional slices. Compared to current 2-D diagnostic mammography with well recognized limitations, KBCT virtually eliminates tissue overlap and superimposition of structures, which are often responsible for failure to diagnose breast cancer early, when it is most treatable, thereby saving lives and reducing ever growing health care costs.
For more information: www.koningcorporation.com


 April 29, 2026
April 29, 2026 









