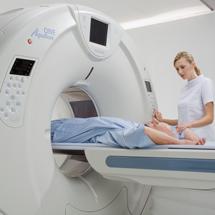
May 21, 2009 – Toshiba America Medical Systems Inc. introduced two advanced multidetector CT systems, the Aquilion Premium edition and the Aquilion CX edition. The Aquilion Premium provides an upgrade path to the Aquilion ONE, while the Aquilion CX is an advanced 64-detector row CT system that incorporates superior features of the existing Aquilion 64.
The Aquilion Premium rounds out the company’s CT portfolio to include a product with 160-detector rows and coverage up to 8 cm in a single rotation. This system is also field-upgradeable to an Aquilion ONE. The Aquilion CX is Toshiba’s next generation 64-detector row CT system featuring faster reconstruction standards with up to 28 images per second, Toshiba’s proven Quantum Advantage detector technology and a patient couch that can accommodate up to 660 lbs. The Aquilion Premium and Aquilion CX are both available now.
Because the Aquilion Premium is field-upgradeable to an Aquilion ONE, this product enables the medical community to have access to advanced technology today, while allowing physicians and administrators to plan for their future needs. Additionally, the Aquilion Premium comes standard with the 72 kW generator, a 660 lb patient weight couch and advanced features like iStation display and 8 cm dynamic scanning capability.
Toshiba’s next generation 64-detector system, the Aquilion CX, is positioned as a comprehensive workhorse CT system and comes standard with faster reconstruction times of 28 images per second, the quickest in the industry. The Aquilion CX is designed for advanced, emergency and routine radiology exams and allows customers to image patients ranging from pediatric to bariatric. The CX features a 72 kW generator for wider mA scanning flexibility, coneXact software for higher definition images for extremities and IACs, SUREExposure, including QDS and Boost3D and double the console storage capacity of a conventional 64-detector CT.
For more information: www.medical.toshiba.com


 April 29, 2026
April 29, 2026 









