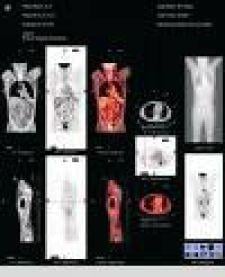
GE Healthcare's Discovery PET/CT 600 is a FDA cleared positron emission tomography/computed tomography (PET/CT) system that is part of GE’s Discovery family of scanners designed to enable earlier detection and accurate monitoring of disease with molecular imaging technology in both hardware and software.
Discovery PET/CT 600 is optimized for use in oncology, which represents more than 90 percent of clinical PET/CT exams. The system leverages the high-speed, high-resolution capabilities of GE’s BrightSpeed CT with the breakthrough motion management capabilities of its Discovery PET system. By combining these scanning technologies, the Discovery PET/CT 600 provides the tools to enable physicians to manage disease with more confidence.
As a key component of the image chain, the Discovery PET/CT 600 includes a scintillator with a proven track record and the highest sensitivity in the industry. GE BGO technology allows for improved lesion detectability, potentially reducing the dose requirement and allowing for faster scans and increased throughput.
For clinicians, the Discovery PET/CT 600 will provide a large 70cm bore that offers a full 70cm PET and CT Field-of-View and a ~225kg (500 lbs.) patient table. The GE Discovery PET/CT 600 offers an increased vertical scan range that provides more flexibility in radiation treatment planning and patient positioning.
“The Discovery PET/CT 600 platform was built for physicians and molecular imaging researchers based on their clinical need for more quantitative accuracy and the power to explore the potential of PET/CT imaging,” said Henry Hummel, general manager of GE Healthcare’s global PET/CT business.


 May 19, 2026
May 19, 2026 









