November 14, 2017 — Spectronic Medical announced that new data for their MRIPlanner software, generating synthetic computed tomography (sCT) images from a single magnetic resonance (MR) image, will be presented in the November issue of the International Journal of Radiation Oncology, Biology, Physics (a.k.a the Red Journal).
The MR-OPERA study shows that an MRI-only treatment planning workflow using the MRIPlanner software is dosimetrically accurate and robust for a variety of equipment vendors, field strengths and treatment techniques. This means that, for the first time, an MRI-only treatment planning workflow for prostate cancer therapy is feasible at most MRI-equipped radiation therapy clinics.
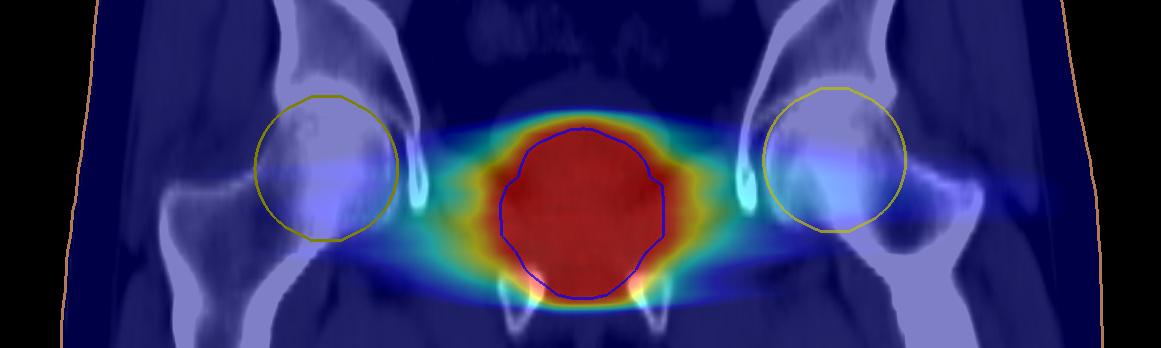
The study also concluded that the differences observed between CT and sCT dose distribution are small, and when compared with other uncertainties in radiation therapy they are negligible.
The overall (multicenter/multivendor) mean dose differences between sCT and CT dose distributions were below 0.25 percent for all evaluated organ and target structures. Gamma evaluation showed a mean pass rate of 99.12 percent (+/- 0.63%, 1 SD) in the complete body volume and 99.97 percent (+/- 0.13%, 1 SD) in the planning target volume using a 2 percent/2-mm gamma criteria.
The results of the study show that the sCT conversion method can be used clinically, with minimal differences between sCT and CT dose distributions for target and relevant organs at risk. The results are consistent between centers, indicating that an MRI-only workflow using MRIPlanner is robust for a variety of field strengths, equipment vendors and treatment techniques.
MRIPlanner is currently CE marked and approved for clinical use in Europe.
For more information: www.spectronic.se


 May 27, 2026
May 27, 2026 









