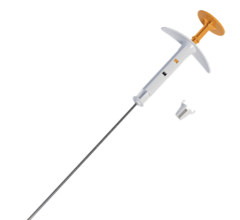
May 20, 2009 - Breast cancer radiation with the SAVI applicator provides positive cosmetic outcomes and the multicatheter device allows greater planning flexibility, technically easier insertion, according to two new studies on breast conservation therapy patients with small breasts, small tumor beds and minimal distance between the lumpectomy cavity and skin surface.
The studies, which were conducted at Moores Cancer Center at University of California, San Diego, will be presented from the podium at the American Brachytherapy Society’s annual meeting, May 31-June 2, 2009, in Toronto.
SAVI is implemented as part of breast conservation therapy, which encompasses surgery to remove the breast cancer and follow-up radiation delivered inside the breast in a procedure known as breast brachytherapy. SAVI treatment is completed in just five days, compared to the six weeks of treatment, five days a week, that are typically required by traditional, external-beam radiation treatment.
The first study was led by Daniel Scanderbeg, Ph.D., and the second study was led by Catheryn Yashar, M.D., associate professor and chief of breast and gynecological services in the UCSD Department of Radiation Oncology.
Dr. Scanderbeg’s study retrospectively evaluated 13 breast cancer patients who chose breast conservation therapy and were successfully treated with SAVI. None of the 13 was eligible for the older form of brachytherapy because they had breast cancer tumor cavities less than 7 mm from the skin surface. That distance is too small for use of the balloon device, because it cannot modulate the dose to minimize skin damage if the cavity is less than 7 mm from the skin surface.
SAVI is not limited by restrictions on skin spacing, because its design enables physicians to direct the radiation dose away from the skin.
According to the study, SAVI provided outstanding coverage of the breast cancer tumor bed while simultaneously sparing normal tissue from radiation. There were no infections, persistent seromas or local recurrences of cancer among any of the patients. Researchers called the cosmetic outcomes “outstanding.”
“SAVI removes the obstacles associated with balloon brachytherapy, namely the inability to customize the radiation dose to avoid damage to critical structures in women with small breasts, small tumor beds, or narrow skin bridges,” said Dr. Scanderbeg. “With this increased flexibility, SAVI allows a greater proportion of women to choose APBI than was previously possible.”
The second study reviewed early clinical experiences with SAVI in 40 breast cancer patients. Nearly half were not candidates for balloon brachytherapy due to skin spacing or small tumor size.
Researchers concluded that the multicatheter device allowed greater planning flexibility, technically easier insertion and minimal exposure to normal tissue.
“As the popularity of APBI has increased, SAVI has allowed us to treat tumor beds as small as 5 cc’s. This device expands the number of women who benefit from accelerated treatment in the form of breast brachytherapy,” said Dr. Yashar.
Patient tolerance among the 40 patients in Yashar’s group was also excellent. There were no symptomatic seromas and the infection rate was 5 percent - lower than published infection rates for balloon brachytherapy.
The SAVI applicator is made by Cianna Medical, Inc. Moores Cancer Center at UC San Diego was the second medical facility in the nation to offer SAVI for breast brachytherapy.
For more information: www.ciannamedical.com


 October 01, 2023
October 01, 2023