December 2, 2020 — Riverain Technologies will showcase its artificial intelligence solutions and enhancements at the now-virtual 2020 Radiological Society of North America Annual Meeting (RSNA).
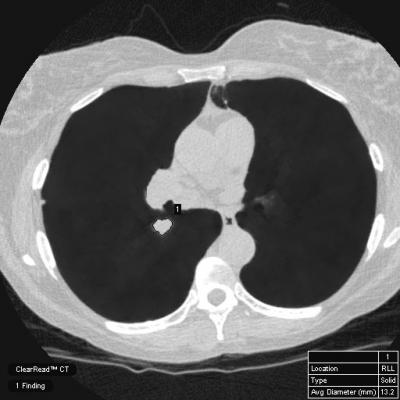
Radiologists spend a significant amount of time on mundane reporting and measurement processes. ClearRead tools help overcome this based on increased automation and can help radiologists improve the efficiency of disease detection, diagnosis, reporting, and treatment.
RSNA visitors will experience how Riverain is harnessing the power of artificial intelligence (AI) and strategic partnerships to empower radiologists, advance radiology, and improve diagnostic imaging.
As part of the event, Riverain will:
- Demonstrate the FDA-cleared and CE-marked ClearRead bone and vessel suppression AI tools that takes advantage of the latest innovations in AI.
- Present a Featured Product Demo on Wednesday, December 2, at 2:00 p.m. CST, followed by a 10-minute Q&A session. RSNA Session Code FD14.
- Present an on-demand Innovation Theater presentation with Thomas Frauenfelder, MD, University Hospital Zurich, as he explains ClearRead benefits and outcomes. RSNA Session Code IT15.
- Present an on-demand AI Showcase presentation with leading thoracic radiologists who have deployed ClearRead in their daily, clinical workflows. They discuss how AI can aid in improving patient outcomes and reading experience and provide their insights on the future of AI in radiology. Guest speakers include Jared Christensen, MD, MBA, Duke Health; Peter Lafferty, MD, LucidHealth; and Terence Matalon, MD, Einstein Health Network. RSNA Session Code AI17.
“Traditionally, radiology has struggled with immense workloads. The challenge continues to accelerate due to more patients and more imaging data per patient. Physician burnout is a recognized issue and will likely get worse. Sites are looking to offset these challenges with AI now,” said Steve Worrell, CEO at Riverain Technologies. “Riverain has expertise in thoracic radiology, partnerships across the industry, and AI-powered solutions to surround radiologists with everything they need to facilitate their reading processes. It’s our privilege to help make sure that every radiologist has the power to see clearly and decide confidently.”
For more information: www.riveraintech.com


 May 12, 2026
May 12, 2026 









