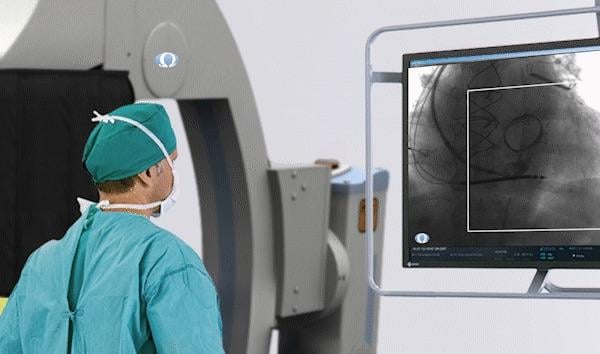
April 4, 2019 — The U.S. Food and Drug Administration (FDA) granted Omega Medical Imaging 510(k) clearance to offer their artificial intelligence (AI)-powered region of interest (ROI) radiation exposure reduction solution FluoroShield for interventional X-ray imaging on their flat panel detector CS-series product lines.
FluoroShield combines AI, an ultra-fast collimator and advanced image processing to deliver an ROI solution that has shown to reduce radiation exposure up to approximately 84 percent. The AI helps minimize X-ray dose to patients and staff while TruBlock technology completely blocks radiation. This delivers maximum radiation reduction while optimizing workflow, helping interventional clinicians better reach their ALARA (as low as reasonably achievable) goals.
For more information: www.omegamedicalimaging.com


 May 04, 2026
May 04, 2026