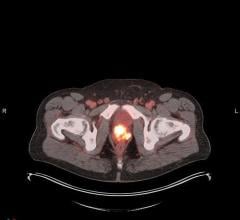
A novel study demonstrates the potential of a novel molecular imaging drug to detect and visualize early prostate cancer in soft tissue, lymph nodes and bone. The research, published in the November issue of The Journal of Nuclear Medicine, compares the biodistribution and tumor uptake kinetics of two Tc-99m labeled ligands, MIP-1404 and MIP-1405, used with SPECT and planar imaging.
Prostate cancer is the most commonly diagnosed non-skin cancer in the United States, and it is second only to lung cancer as the leading cause of cancer deaths in American men. An estimated 233,000 new cases of prostate cancer will be diagnosed in the United States in 2014, and an estimated 29,000 will die of the disease. More than 2 million men are currently living with prostate cancer in the United States.
Under an exploratory investigational new drug, using a cross-over design, researchers compared the pharmacokinetics, biodistribution, and tumor uptake of Tc-99m MIP-1404 and Tc-99m MIP-1405 in 6 healthy men and 6 men with radiographic evidence of metastatic prostate cancer. Whole body images were obtained at 10 minutes and at 1, 2, 4 and 24 hours. SPECT was performed between 3 and 4 hours after injection. Prior to the study, no single target-specific Tc-99m radiopharmaceutical could image prostate cancer in soft tissue, lymph nodes and bone (bone metastasis) based on planar and SPECT. There was no uptake in degenerative bone disease, which often confounds bone scans.
“This research represents an innovative prostate cancer planar and SPECT imaging technology—addressing unmet clinical need for sensitive and selective imaging of loco-regional and distant metastatic prostate cancer,” stated Shankar Vallabhajosula, Ph.D., lead author of the study “99mTc-Labeled Small Molecule Inhibitors of Prostate Specific Membrane Antigen: Pharmacokinetics and Biodistribution Studies in Healthy Subjects and Patients with Metastatic Prostate Cancer. “With respect to imaging, the lack of focal uptake in the normal prostate of healthy volunteers with both compounds further demonstrated that PSMA is a viable targeting mechanism for detection and visualization of prostate cancer and suggests that this imaging approach is highly sensitive and disease specific.”
There was good correlation with bone scans in most subjects, although in general, more lesions were visualized with MIP-1404 and MIP-1405 than with bone scans, suggesting this agent may be more sensitive to detecting skeletal or marrow invasion earlier than bone scans. “We also demonstrated that Tc-99m MIP-1404 has favourable pharmacokinetics and biodistribution, which represents a breakthrough in imaging of prostate cancer for the following reasons: Tc-99m MIP-1404 can image prostate cancer in lymph nodes, soft tissue and bone,” noted Vallabhajosula.
A multi-center phase II study with Tc-99m MIP-1404 in 100 patients was recently completed, and the data were presented at 2014 SNMMI Annual Meeting in St. Louis, Mo. Progenics Pharmaceuticals has plans to conduct a phase III trial soon.
Authors of the article “99mTc-Labeled Small Molecule Inhibitors of Prostate Specific Membrane Antigen: Pharmacokinetics and Biodistribution Studies in Healthy Subjects and Patients with Metastatic Prostate Cancer” include Shankar Vallabhajosula, Anastasia Nikolopoulou, Joseph Osborne, Scott T. Tagawa, Irina Lipai, Lilja Solnes, and Stanley Goldsmith, New York Presbyterian Hospital and Weill Cornell Medical College, New York, N.Y.; John Babich, Kevin P. Maresca, Thomas Armor, John Joyal, and Robert Crummet, Molecular Insight Pharmaceuticals, Inc., Cambridge, Mass.; and James B. Stubbs, Radiation Dosimetry Systems, Inc., Alpharetta, Ga.
For more information: http://jnm.snmjournals.org


 April 07, 2026
April 07, 2026