February 15, 2019 – University of Minnesota Health (M Health) is the first in the United States to begin offering GammaTile Therapy, a new approach to treating recurrent brain tumors. GammaTile Therapy is a U.S. Food and Drug Administration (FDA)-cleared, surgically targeted radiation therapy (STaRT) manufactured by GT Medical Technologies that is designed to delay tumor regrowth for patients with brain tumors.
The first patient was treated by University of Minnesota physician Clark C. Chen, M.D., Ph.D., head of the Department of Neurosurgery at the University of Minnesota Medical School.
“At University of Minnesota Health, our mission is to advance new, safe and effective therapeutic options for the many brain tumor patients who did not respond to the standard-of-care therapies,” Chen said. “Moreover, the University of Minnesota Medical School’s Department of Neurosurgery has a long-standing history of contribution in radio-biologics. To be the first institution in the U.S. to offer the GammaTile Therapy is particularly satisfying in this context.”
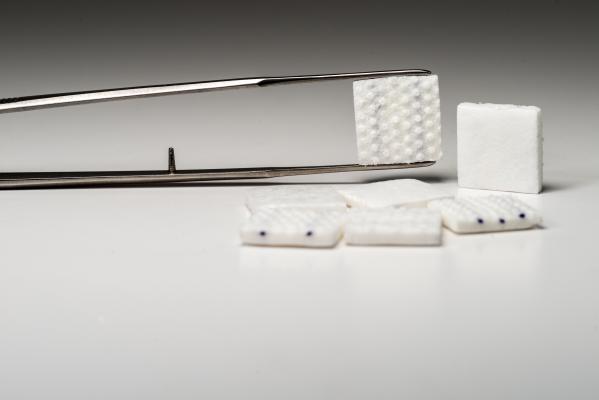
Aggressive brain tumors tend to be resistant to current treatments and nearly always recur. Outcomes for patients with brain tumors have improved very little over the past 30 years. GammaTile is FDA-cleared for patients with recurrent brain tumors. It consists of a bioresorbable, conformable 3-D-collagen tile embedded with a Cesium radiation source. GammaTile is placed at the time of surgery so that it immediately begins to target residual tumor cells with radiation while limiting the impact on healthy brain tissue.
“I am optimistic that GammaTile will impact the clinical outcome for our brain tumor patients, particularly when combined with appropriate medical therapy,” explained Chen.
GammaTile Therapy offers some advantages over other treatments for patients undergoing surgery for recurrent brain tumors. A course of external beam radiation therapy (EBRT), for example, requires daily treatments for up to six weeks; in contrast, patients treated with GammaTile Therapy require no additional trips to the hospital or clinic. Additionally, many patients may not be candidates for EBRT at the time of tumor recurrence because the risk of additional EBRT outweighs the potential benefits. Finally, those patients who may be candidates for EBRT typically have to wait four weeks or more for surgical wound healing before beginning treatment, allowing residual, microscopic tumors to grow during this waiting period.
Chen has conducted research that supports the efficacy of radiation treatment immediately after resection. Published in the Journal of Neuro-Oncology, Chen’s study showed that patients with glioblastoma, the most common form of primary brain cancer in adults who received immediate postoperative radiation exhibited improved survival relative to those who did not.
“I am pleased to be able to offer a more targeted radiation therapy to my patients,” said Kathryn E. Dusenbery, M.D., head of the Department of Radiation Oncology at the University of Minnesota Medical School. “With GammaTile, we apply radiation therapy exactly where it is needed, without harming surrounding tissue, and patients do not need to come back for ongoing radiation treatments. In addition, this new targeted approach may help reduce the burden of ongoing radiation treatment and help my patients and their caregivers experience a better quality of life.”
Additional data supporting the efficacy and safety profile of the therapy for patients with recurrent, previously treated meningiomas were published last month in the Journal of Neurosurgery (JNS), the official journal of the American Association of Neurological Surgeons (AANS). Clinical data from other types of tumors will be presented at the AANS Annual Scientific Meeting, April 13-17 in San Diego.
For more information: www.gtmedtech.com


 May 06, 2026
May 06, 2026 









