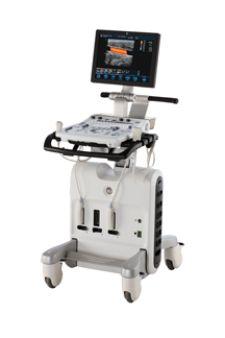
GE Healthcare launched its new mobile Vivid S5 cardiovascular ultrasound system, created for multiple care areas, featuring a full-sized 17-inch LCD monitor, three active transducer connectors and room for on-board peripherals retained in a miniaturized console weighing less than 160 pounds.
Inspired by the Vivid i, Vivid 7 and Vivid 4, this new concept encompasses stress echo capabilities and raw data DICOM to expand the reach of patient examinations. The system also features backward data compatibility with the rest of GE Healthcare’s Vivid family, including Vivid 3 and Vivid 4, as well as enhanced connectivity and productivity tools and a new ergonomic design.
The “Flex-Fit” mechanism provides constant pivoting height adjustment of the control panel, while reportedly keeping optimal distance from the user and leaving legroom for standing or sitting positions. Its horizontal keyboard swivel works to ensure optimal placement and comfort in operation.


 May 07, 2026
May 07, 2026 









