January 12, 2024 — NeuroLogica Corp. announced its OmniTom Elite PCD has received an Innovative Technology designation from Vizient, Inc., the largest provider-driven healthcare performance improvement company in the country. NeuroLogica Corp. exhibited OmniTom Elite PCD at the Vizient Innovative Technology Exchange on Oct. 17 in Dallas.
Each year, healthcare experts serving on Vizient customer-led councils review select products and technologies for their potential to enhance clinical care, patient safety, healthcare worker safety or to improve business operations of healthcare organizations. Innovative Technology designations are awarded to previously contracted products to signal to healthcare providers the impact of these innovations on patient care and the business models of healthcare organizations.
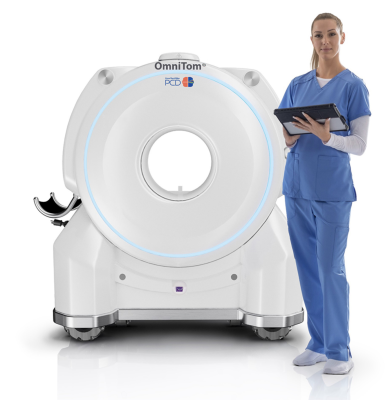
NeuroLogica’s OmniTom Elite PCD is a groundbreaking innovation in medical imaging. This mobile CT scanner boasts high-resolution imaging and material decomposition capabilities, enabling healthcare professionals to conduct precise neuroimaging at the patient's bedside in the ICU, or in another area designated by the facility such as the ER department. Cleared for use on adults, the reduction of artifacts can increase the ability to identify a bleed along the skull vs contrast, reducing the risk of a potential misdiagnosis, and helping to better identify pathology in the posterior fossa. Post thrombectomy, it can differentiate between blood and iodine staining without needing to wait. Where conventional CT PCD, Dual Energy, or Spectral CT imaging is in use, the NeuroLogica OmniTom PCD scanner can help to standardize imaging system-wide.
“NeuroLogica has always been driven to innovate and expand the world of healthcare,” said Renaud Maloberti, Head of mCT Business. “From the creation of mobile CT to implementation of the first single source, single detector photon counting CT, we are witnessing the next stage of imaging care and standardization.”
“Congratulations to NeuroLogica Corp. for achieving this peer-designated status,” said Kelly Flaharty, Vizient senior director of contract services. “Our customer-led council deemed OmniTom Elite PCD worthy of this designation for its potential to make an incremental difference in healthcare.”
Vizient represents a diverse customer base that includes academic medical centers, pediatric facilities, community hospitals, integrated health delivery networks and non-acute healthcare providers and represents approximately $130 billion in annual purchasing volume. Through its Innovative Technology Program, Vizient works with customer-led councils and task forces to evaluate products for their potential to bring real innovation to healthcare. Vizient may award a contract to products deemed worthy of the Innovative Technology designation outside of the competitive bid cycle.
For more information: https://www.neurologica.com/


 May 20, 2026
May 20, 2026 









