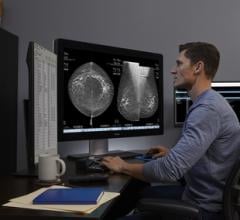
DIAGNUM Workstation for mammography by FENICS.
October 13, 2009 - FENICS has validated the Matrox Xenia PRO display controller board with their DIAGNUM Workstation mammography computer-aided detection (CAD) solutions, which will be demonstrating this technology at Journee Française de Radiologie (JFR) 2009 from October 16-20, 2009, in Paris - Porte Maillot, France.
FENICS is the first company from France to propose a set of aided diagnosis solutions for mammography through its DIAGNUM product line. Its DIAGNUM Look solution is an interactive CAD that detects anomalies that also routes and characterizes them to generate reports in the current language. With its strong performance and user friendly interface, the workstation has already attracted the attention of many radiologists.
Matrox Xenia a native PCI Express single-slot board with all-digital, triple-monitor output, and the flexibility that drives display configurations. Xenia Series supports resolutions from under 1MP to 8MP, with up to 1GB of on-board memory, and features new technology to ensure optimum display calibration (via Matrox DLC and 8/10/13-bit gamma LUTs).
Xenia Series is an expandable solution, that provides medical imaging developers and integrators to increase hardware-accelerated performance via hardware LUTs, multiple hardware window IDs, and additional hardware features.
For more information: www.matrox.com/graphics and www.fenics-sas.com


 April 20, 2026
April 20, 2026