March 23, 2018 — Materialise NV became the first company in the world to receive U.S. Food and Drug Administration (FDA) clearance for software intended for 3-D printing anatomical models for diagnostic use. The company said leading hospitals are adopting integrated 3-D printing services as part of their medical practices as they recognize the added value it brings to personalized patient care.
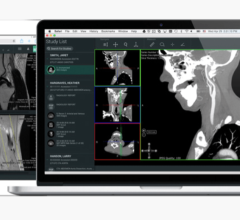
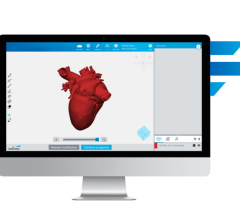
In August 2017, the FDA announced that software intended to create output files used for printing 3-D patient-specific anatomical models used for diagnostic purposes is a Class II medical device and requires regulatory clearance. Materialise is the first company to provide software that conforms to these regulations and that can be used in U.S. hospitals in combination with a compatible 3-D printer. Materialise Mimics inPrint software is used for pre-operative planning and the fabrication of physical models for diagnostic purposes, including patient management, treatment and surgeon-to-surgeon communication.
Frank J. Rybicki, M.D., Ph.D., chief of medical imaging at Ottawa Hospital, Ontario, Canada, said: “510k clearance is an essential component to ensure quality and safety in the practice of anatomical modeling in hospitals. This milestone for Materialise serves as a benchmark for the clinical implementation of 3-D printing for physicians creating 3D models at the point-of-care.”
The FDA clearance supports the creation of point-of-care 3-D printing facilities in hospitals. Anatomical models help surgeons to make better-informed decisions and to accurately plan their surgeries. They are also used to enhance education and communication between multidisciplinary teams and with the patient. Materialise said an increasing number of hospitals across the U.S. have integrated the practice of 3-D printing in their medical care and are creating point-of-care 3-D printing facilities. Out of the top 20 U.S. hospitals (as ranked by the U.S. News and World Report), 16 have reportedly implemented a medical 3-D printing strategy using Materialise Mimics technology.
For more information: www.materialise.com


 April 18, 2025
April 18, 2025