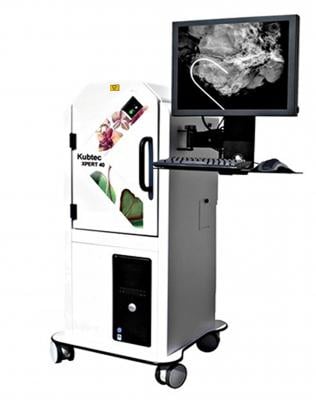
Kubtec announced the U.S. Patent & Trademark Office has issued Patent No. 14/503,130 for the technology incorporated into the Mozart System with TomoSpec Technology. This patent recognizes that the Mozart System with TomoSpec Technology, the world’s first portable 3-D Digital Specimen Tomosynthesis Imaging System, is unique in its field.
The Mozart System with TomoSpec Technology allows surgeons to bring 3-D tomosynthesis imaging directly into the Operating Room for lumpectomy and partial mastectomy procedures. It gives surgical teams the ability to make faster and more precise intraoperative determinations of the successful excision of tumor margins. Prior to the Mozart System with TomoSpec Technology, surgeons were restricted to less effective modes of multiple 2-D imaging, or waiting while a specimen was transported from the operating room to another location for 3-D analysis.
“Surgeons often do not have the time to wait for off-site 3-D image analysis. A result of this is that around 25 percent of lumpectomy patients will require a second procedure1, as follow up analysis frequently indicates that the tumor has not been completely excised during surgery,” said Vikram Butani, president and CEO of Kubtec. “Our goal is to reduce that number by giving surgeons and radiologists the most advanced digital specimen radiography tools that enable them to precisely identify tumor margins during the operation itself. This shortens procedure times and decreases the risk that a re-excision will be necessary. That’s better for the patient and it reduces hospital costs, too.”
Chester Lowe, Ph.D. director of research and development at Kubtec, commented, “This patent confirms how unique and important the technologies integrated into the design of the Mozart Digital Specimen Tomosynthesis Imaging System really are. We believe that the Mozart System with TomoSpec Technology will have a major impact on breast cancer procedures and drive positive outcomes for patients.”
For more information: www.kubtec.com


 April 20, 2026
April 20, 2026 









