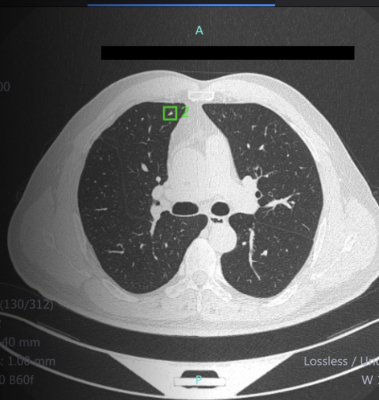
July 10, 2020 — Infervision announced U.S. Food and Drug Administration (FDA) 510(K) clearance of the InferRead Lung CT.AI product, which uses the state-of-the-art artificial intelligence and deep learning technology to automatically perform lung segmentation, along with accurately identifying and labeling nodules of different types. InferRead Lung CT.AI is designed to support concurrent reading and can aid radiologists in pulmonary nodule detection during the review of chest computed tomography (CT) scans, increasing accuracy and efficiency. With five years of international clinical use, Infervision's InferRead Lung CT.AI application is a robust and powerful tool to assist the radiologist.
InferRead Lung CT.AI is currently in use at over 380 hospitals and imaging centers globally. More than 55,000 cases daily are being processed by the system and over 19 million patients have already benefited from this advanced AI technology. "Fast, workflow friendly, and accurate are the three key areas we have emphasized during product development. We're very excited to be able to make our InferRead Lung CT.AI solution available to the North American market. Our clients tell us it has great potential to help provide improved outcomes for providers and patients alike," said Matt Deng, Ph.D., Director of Infervision North America. The Company offers the system under a number of pricing models to make it easy to acquire.
The company predicts the system may also be of great benefit to lung cancer screening (LCS) programs across the nation. Lung cancer is the second most common cancer in both men and women in the U.S. Survival rates are 60% in five years if discovered at an early stage. However, the survival rate is lower than 10% if the disease progresses to later stages without timely follow-up and treatment. The Lung Cancer Screening program has been designed to encourage the early diagnosis and treatment of the high-risk population meeting certain criteria. The screening process involves Low-dose CT (LDCT) scans to determine any presence of lung nodules or early-stage lung disease. However small nodules can be very difficult to detect and missed diagnoses are not uncommon.
"The tremendous potential for lung cancer screening to reduce mortality in the U.S. is very much unrealized due to a combination of reasons. Based on our experience reviewing the algorithm for the past several months and my observations of its extensive use and testing internationally, I believe that Infervision's InferRead Lung CT.AI application can serve as a robust lung nodule "spell-checker" with the potential to improve diagnostic accuracy, reduce reading times, and integrate with the image review workflow," said Eliot Siegel, M.D., Professor and Vice Chair of research information systems in radiology at the University of Maryland School of Medicine.
InferRead Lung CT.AI is now FDA cleared, and has also received the CE mark in Europe. "This is the first FDA clearance for our deep-learning-based chest CT algorithm and it will lead the way to better integration of advanced A.I. solutions to help the healthcare clinical workflow in the region," according to Deng. "This marks a great start in the North American market, and we are expecting to provide more high-performance AI tools in the near future."
For more information: global.infervision.com


 May 22, 2026
May 22, 2026 









