Photo: Elucid
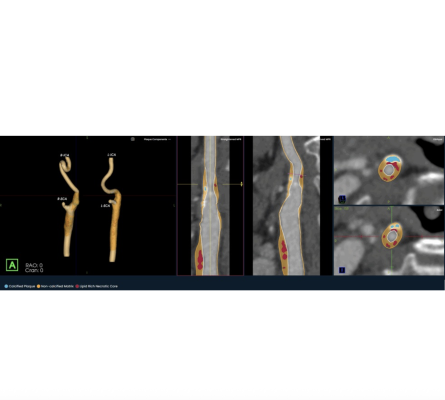
Oct., 2025 — Elucid, an AI medical technology company focused on providing physicians with a more precise view of atherosclerosis, has introduced its PlaqueIQ image analysis software for the quantification and classification of plaque morphology in the carotid arteries. The first and only CT-based plaque analysis software indicated for the carotid vasculature, PlaqueIQ may help physicians diagnose carotid plaques at risk for rupture and develop patient-specific treatment pathways to prevent and monitor against ischemic stroke.
“The ability to characterize plaque composition precisely revolutionizes carotid stroke risk assessment,” said Dr. David Deaton, surgeon and vascular specialist; adjunct associate professor of Surgery, Perelman School of Medicine, University of Pennsylvania. “Plaque composition has long been recognized as a critical risk element for stroke but was not available with conventional imaging techniques. Precise determination of plaque composition supersedes the traditional stenosis assessment and allows a more accurate identification of patients at highest risk for carotid stroke and facilitates procedural choice and planning.”
“For years, we’ve used CT to quantify coronary plaque and personalize prevention and, now, being able to do the same with the carotid arteries is a game-changer,” said Dr. Alberto Morales, founder of South Tampa Cardiology. “This technology finally lets us measure and monitor carotid plaque biology, not just anatomy, giving us a much more complete picture of stroke risk and overall vascular health. This is truly the future of vascular medicine.”
PlaqueIQ is the first and only FDA-cleared, non-invasive plaque analysis based on objective histology, offering physicians non-invasive CT Virtual Histology. PlaqueIQ delivers quantification and classification of plaque morphology and is uniquely capable of identifying and quantifying lipid rich necrotic core, the plaque type strongly linked with both cardiovascular and cerebrovascular risk.2,3,4
PlaqueIQ is designed to help physicians prioritize and personalize treatment based on actual coronary and carotid artery disease, rather than population-based risk. The technology supports physicians in evaluating treatment of patients’ symptoms and risk of future events, such as heart attack and stroke, and enables the development of personalized care pathways informed by each patient’s individual plaque characteristics.
When used for both the coronary and carotid arteries, PlaqueIQ non-invasively delivers a quantitative and qualitative assessment of systemic atherosclerotic risk and creates the potential to perform both coronary and carotid plaque analysis in a single scan.
“At Elucid, it’s our mission to go after the number one source of heart attack and stroke, and that is plaque wherever it occurs in the coronary or carotid vasculature,” said Elucid’s CEO Kelly Huang, PhD. “We envision a day that with just one scan, physicians can gain a powerful understanding of systemic atherosclerotic risk in order to personalize treatment for and monitor the whole patient.”
For additional information, please visit elucid.com.
References
- Lal, Brajesh K., et al. Computed tomography angiographic biomarkers help identify vulnerable carotid artery plaque. Journal of Vascular Surgery. April 2022. 75(4). 1311-1322.e3. Available from www.sciencedirect.com/science/article/pii/S0741521421024435.
- Virmani, Renu, et. al. Lessons From Sudden Coronary Death : A Comprehensive Morphological Classification Scheme for Atherosclerotic Lesions. Arteriosclerosis, Thrombosis, and Vascular Biology. May 2020. 20(5). Available from www.ahajournals.org/doi/10.1161/01.ATV.20.5.1262.
- Sakamoto, Atsushi, et. al. Vulnerable Plaque in Patients with Acute Coronary Syndrome: Identification, Importance, and Management. US Cardiology Review. Jan. 2022. 26:16:e01. Available from www.uscjournal.com/articles/vulnerable-plaque-patients-acute-coronary-syndrome-identification-importance-and
- Gupta, Ajay, et. al. Carotid Plaque MRI and Stroke Risk: A Systematic Review and Meta-analysis. Stroke. Aug. 2013. 44(11). Available from www.ahajournals.org/doi/10.1161/strokeaha.113.002551.


 May 26, 2026
May 26, 2026 









