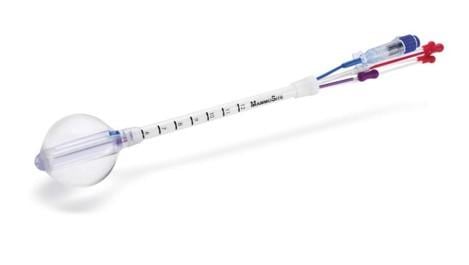
Should cancer be detected in its early stages, many women are candidates for Hologic's targeted radiation therapy, a patient-friendly and effective alternative for women that qualify. At RSNA 2011, Hologic will feature its next generation MammoSite multi-lumen (ML) brachytherapy applicator.
For women with early-stage breast cancer, Hologic offers today’s most widely used form of partial breast irradiation: MammoSite five-day targeted radiation therapy. MammoSite therapy gives physicians an important tool for the practice of breast-conserving therapy, and it makes it easier for more women to consider the choice of lumpectomy, including some women who may have otherwise had a mastectomy to avoid lengthy conventional radiation therapy. Hologic continues to expand the horizon of Accelerated Partial Breast Irradiation (APBI) for early-stage breast cancer patients with its next generation MammoSite multi-lumen (ML) brachytherapy applicator. The MammoSite ML technology enables the ability to shift the radiation dose for a more precise target. This feature will result in more women being appropriate candidates for APBI.
For more information: www.hologic.com


 May 06, 2026
May 06, 2026