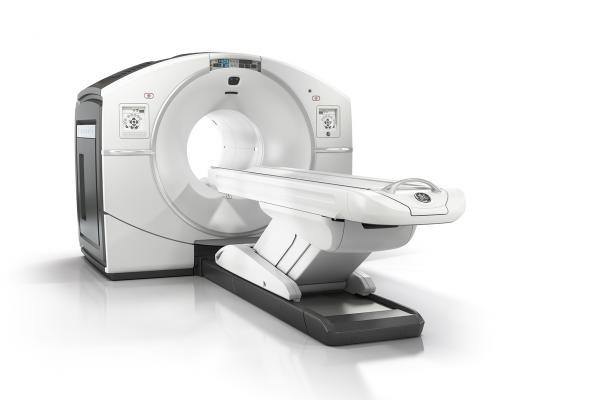
September 24, 2014 — GE Healthcare announced U.S. Food and Drug Administration (FDA) 510(k) clearance of its Discovery IQ PET/CT (positron emission tomography/computed tomography) system.
Physicians not only want the ability to detect smaller lesions, but also the ability to determine whether the patient is responding to current treatment. Discovery IQ delivers the highest NEMA (National Electrical Manufacturers Association) sensitivity (up to 22 cps/kBq) and the largest axial field-of-view (up to 26 cm) compared to other PET/CT equipment. With Q.Clear it is designed to provide more accurate quantitation (SUVmean) with high signal-to-noise ratio (SNR) for small lesion detection, fast and efficient reading, and a confident diagnosis.
“By 2020, it’s estimated that 50 percent of people will develop cancer at some point in their lives and we also know that currently, approximately 70 percent of cancer patients do not respond to their initial chemotherapy treatment,” said Wei Shen, general manager of GE Healthcare PET/CT. “I’m excited about the recent FDA clearance of Discovery IQ, which will help physicians achieve their primary mission of delivering the best possible patient outcomes. And, by making Discovery IQ mobile-ready, we engineered it to be accessible to more patients in more places, allowing for high-performance PET/CT clinical care to whoever needs it.”
GE Healthcare’s new Q.Clear technology is a critical component of Discovery IQ. It delivers, for the first time, no trade-off between image quality and quantitative SUV measurements. By providing two times improvement in both quantitative accuracy (SUVmean) and image quality (SNR) in PET/CT imaging, this new tool provides benefits to physicians across the cancer care continuum from diagnosis and staging to treatment planning and assessment.
For more information: www.gehealthcare.com


 April 23, 2026
April 23, 2026 









