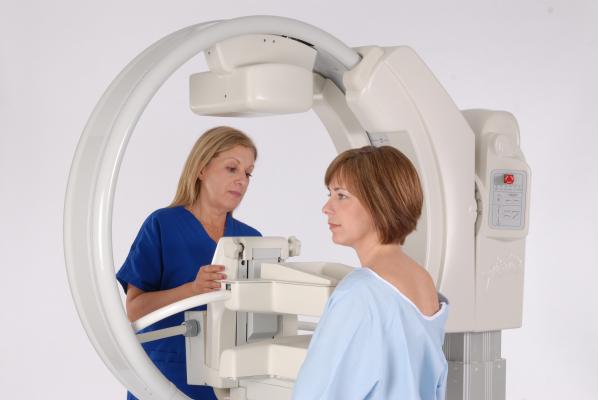
October 14, 2015 — Gamma Medica Inc. announced the CE Mark approval allowing for the sale and distribution of the LumaGEM Molecular Breast Imaging (MBI) system within the European Economic Area (EEA). Gamma Medica is finalizing distribution partners in strategic markets across the EEA.
Breast cancer is the most common cancer affecting women in Europe, with nearly one in 10 women developing breast cancer before the age of 80. Dense breast tissue has been linked to an increased risk of breast cancer and a decrease in mammography sensitivity. These factors impact early detection efforts as both cancer and dense tissue appear white on the mammogram.
The LumaGEM MBI system addresses the critical shortcomings of mammography and other screening modalities in the early detection of breast cancer for the 50 percent of women with dense breast tissue. The system has been commercially available in the United States since obtaining approval by the U.S. Food and Drug Administration (FDA) in 2011.
A recent study conducted by Mayo Clinic, and published in two articles in the American Journal of Roentgenology (AJR) in 2015 determined that the addition of MBI, such as the LumaGEM MBI system, to an annual mammogram screening demonstrated a near 400 percent increase in invasive breast cancer detection rates. The combination also demonstrated a 50 percent reduction in biopsies and a subsequent 15 percent lower cost per cancer detected than screening with mammography alone.
The growing awareness of the limitations of mammography screening for women with dense breast tissue and the necessity for an effective adjunct screening technology led to the creation of LumaGEM MBI. Unlike mammography, tomosynthesis, ultrasound and magnetic resonance imaging (MRI) — all of which are anatomical imaging tests — MBI is a functional imaging modality that highlights metabolic activity in the breast and detects tumors not visible on a mammogram due to tissue density.
It is important for women to discuss with their doctors if MBI is an appropriate screening method for them. In addition to women with dense breast tissue or suspicious lesions, MBI may also be valuable for high-risk patients with a family history of breast cancer, patients who have tested positive for BRCA1 or BRCA2 genes, symptomatic patients, patients with cosmetic augmentation or patients who cannot receive an MRI.
For more information: www.gammamedica.com


 May 22, 2026
May 22, 2026 









